INJECTION
Interview: Celebrating 10 Years of Molly Enabling Patients' Independence
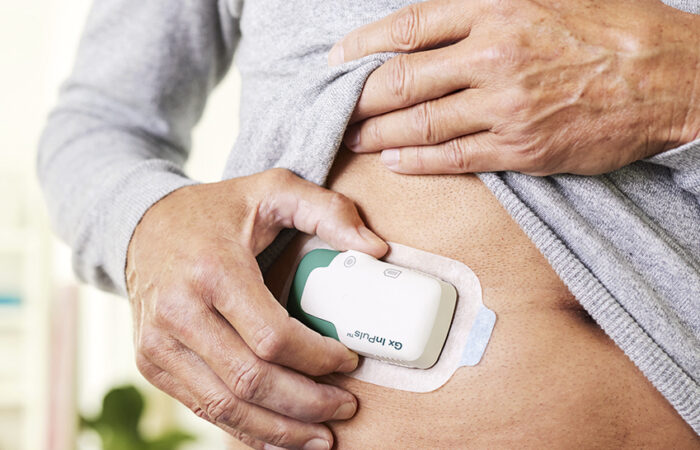
Nicholas Heaton looks back on the decade since Molly – SHL Medical’s signature two-step autoinjector platform – received its first approval, reviewing what made the device platform a success, where Molly is positioned in the market today and how it will continue to evolve to meet the demands of the future.
8th Sep 2026 - 10th Sep 2026
Parma, Province of Parma, Italy
10th Jun 2026 - 10th Jun 2026
London, UK