To Issue 134
Citation: Kaiser-Kershaw S, “How Recent NFC Advances Enable Anti-Counterfeit Drug Containment and Support Adherence”. ONdrugDelivery, Issue 134 (Jun 2022), pp 30–33.
Sylvia Kaiser-Kershaw discusses how recent near-field communication tagging advancements can enable pharmaceutical companies to protect the authenticity and integrity of their products, while also making it easier for healthcare professionals and patients to use medications more effectively through intelligent drug delivery systems.
“Some of the latest NFC smart tags come with a SUN authentication message for web authentication, which changes dynamically upon each scan or “tap”, allowing pharma companies to combat counterfeits and supply chain fraud more effectively.”
THE RISE OF COUNTERFEIT DRUGS
Today’s pharma industry faces an ever increasing array of challenges, including rising numbers of counterfeit products, attacks on its supply chains, adverse conditions affecting the integrity of its products and how to ensure customer loyalty. The WHO estimates that around 10% of all medicines sold worldwide are counterfeit.1 The latest intellectual property crime threat assessment report, produced jointly between Europol and the EU Intellectual Property Office (EUIPO), found that an increasing number of counterfeit pharmaceutical products have been identified in recent years.2
The typical targets for pharmaceutical counterfeiting include expensive drugs, such as vaccines, cancer medicines and antibiotics; however, a significant number of counterfeit high-demand products, such as face masks and hand sanitisers, were also identified during the covid-19 pandemic. As supply chain complexity, from production to packaging, distribution and sales across different markets, and the prevalence of e-commerce, such as online pharmacies, increase throughout the pharma industry, this counterfeiting problem is becoming more widespread.
CONVENTIONAL ANTI-COUNTERFEITING METHODS
Pharma companies have tried numerous ways to fight drug counterfeiting by using item-level serialisation and optical codes, such as data matrix and QR codes. These coding systems are static and relatively easy to copy, which limits their ability to prevent counterfeiting. The data matrix system is based on serialisation legislation, providing traceability throughout the distribution supply chain until the point of dispensation but does not include the last mile to the patient. Additionally, optical devices lack the functionality to operate as advanced “sensor platforms” – they cannot register external events, such as tampering or a change in fill level or temperature.
“Innovative NFC tags with a capacitive structure can also be used as passive sensing devices to detect changes in a condition, such as moisture or fill level, without a battery.”
NFC FOR AUTHENTICATION AND TAMPER-PROOFING
Near-field communication (NFC) smart tags can be read by any NFC-enabled device. Globally, there are about 3.4 billion NFC enabled devices, primarily smartphones, in use today.3 This form of authentication does not require a costly specialised reader device and can be done by anyone involved in the authentication and tracking process.
Beyond a unique identifier, NFC authentication tags come with additional electronic security attributes. Some of the latest NFC smart tags come with a secure unique NFC (SUN) authentication message for web authentication, which changes dynamically upon each scan or “tap”, allowing pharma companies to combat counterfeits and supply chain fraud more effectively. By adding a unique ID, tap counter and status value to the programmed NFC message, along with a cryptographic message authentication code, only an original tag can generate a valid SUN message, and each tagged item can be reliably authenticated, while its digitised status value is protected against malicious changes.
When using NFC authentication, counterfeits are easy to spot because brand inspectors, pharmacists and consumers can all verify product authenticity using their smartphones without downloading a special app (Figure 1). Patients can even become more deeply engaged with their medication supplier by taking advantage of on-demand access to product information and dosage instructions, including videos, helpful digital tools and more.

Figure 1: Counterfeits are easy to spot because brand inspectors, pharmacists and consumers can verify product authenticity with their smartphones.
A unique tag ID can be assigned to a specific distributor and location, reporting their origin and intended destination at any point in the supply chain. In addition, some tags offer a mutual authentication option with a cryptographic key, ensuring that only an authorised reader or server can access the tag’s data. The tag only releases data to an authenticated reader when the tag is confirmed, protecting sensitive tag data against unauthorised access and/or malicious change attempts in the supply chain.
Electronically tamper-evident NFC conductive seals and labels can be attached to drug packaging at the point of manufacture to protect against unauthorised opening. When the conductive loop in the seal breaks, it irreversibly writes the “once opened” status into the tag memory and sends the opened status to the cloud with the tap of a phone. In comparison, capacitive tamper-proof tags measure capacitance and compare it with preconfigured limits upon readout with an NFC-enabled smartphone. When these limits are exceeded, the open status is added to the SUN message. This mode of tamper detection is difficult for fraudsters to reconstruct and is suitable for integration into physical form factors such as bottle closures. Both tamper-evident modes work with web-based authentication.
Innovative NFC tags with a capacitive structure can also be used as passive sensing devices to detect changes in a condition, such as moisture or fill level, without a battery. Here, if values are outside the predefined range, changes in capacitance are captured by the NFC device and then interpreted by a cloud or smartphone application.
“When attached to a drug delivery device, such as an autoinjector or inhaler, NFC tags can aid patients with medication adherence.”
NFC FOR MANAGING CHRONIC CONDITIONS
Beyond counterfeit and illicit drugs, the healthcare industry and, in particular, pharma companies are facing another challenge, which is the global increase in chronic illnesses and how best to treat them. In 2019, the Global Burden of Disease (GBD) study reported that chronic conditions now account for millions of deaths worldwide each year.4
While metabolic conditions, such as obesity, diabetes, hypertension and high cholesterol, have the most significant impact on health, other conditions, such as asthma, arthritis and mental disorders, are also on the rise. Treatments for these illnesses typically require long-term use of medications, and it is not always easy for patients to stay on track. Not getting their prescription filled on time or forgetting to take their medication are just two common ways patients can struggle to stick to their prescribed dosing regimen. For patients taking multiple medications for a range of conditions, there can also be an issue of not understanding the directions for proper dosing or uncertainty of when to take each medication, also known as medication adherence.
According to the WHO, patient adherence to long-term medication therapy in developed countries averages 50% of patients, with even lower rates in developing countries.5 The consequences of lapses in medication adherence can lead to poor health outcomes for patients and increased healthcare costs. This is a challenge NFC tags can help with.
When attached to a drug delivery device, such as an autoinjector or inhaler, NFC tags can aid patients with medication adherence (Figure 2). By simply tapping a tagged device with a smartphone, a link can be created to medication guides and usage videos, including advice on possible side effects and other helpful information. Patients can also set up daily reminders for their treatments by downloading a specific app.
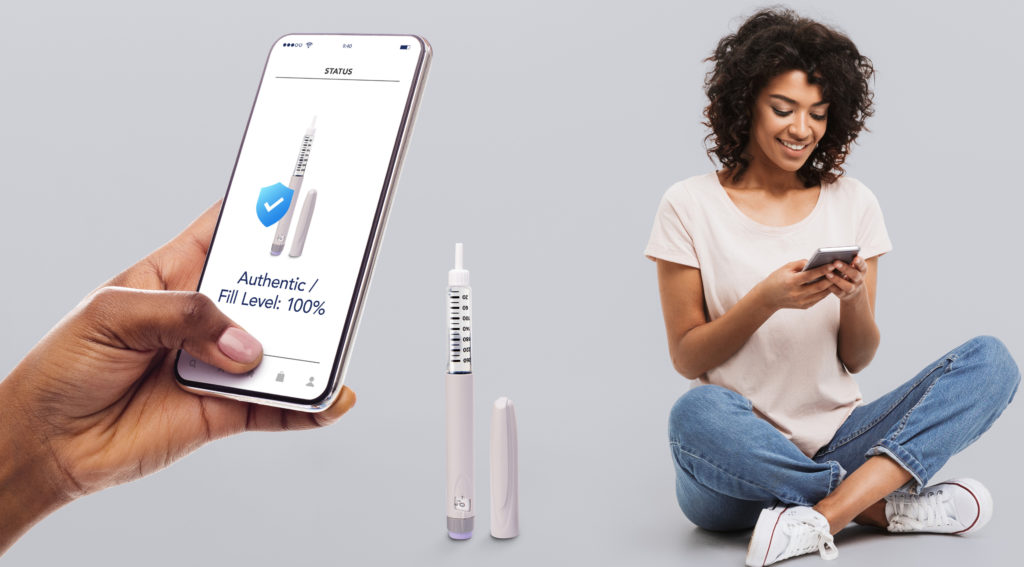
Figure 2: When attached to a drug delivery device, such as an autoinjector or inhaler, NFC tags can help with medication adherence.
For multi-use refillable drug delivery devices, adding an NFC reader can help in additional ways. When built into the drug delivery device, the reader can authenticate the NFC tag on a prefilled drug consumable, such as a cartridge, confirming that it is from a trusted source. If the NFC tag is encoded with the drug’s expiration date, the reader on the device can also confirm that it remains viable. In addition, the reader can record that the medication has been taken after each dose, and the tag can even store the incremental counter value. Adding Bluetooth connectivity to the drug delivery device can ensure that this data syncs with the patient’s mobile app. This data can also be stored securely in the cloud so that, with a patient’s permission, their healthcare practitioner can track their adherence history and provide personalised feedback (Figure 3).

Figure 3: With a patient’s permission, data about their adherence can be stored securely in the cloud, enabling a healthcare practitioner to track their adherence history and provide personalised feedback.
Advanced battery-free capacitive sensing NFC technologies can also detect fill levels. Smart injection devices can verify the dosage level when tapped with an NFC phone with the right app to ensure that the medication has been taken correctly. This technology can also be used to check the fill level of medicines stored in opaque packaging and send the patient a reminder, via an app, to replenish their prescriptions when the level is below a predetermined threshold. NFC tags with an integrated sensor and micro controller can also support smart temperature measurements; for example, an insulin injection pen could signal when the dose is at room temperature, making the injection less painful for the user.
CONCLUSION
Pharma companies play an essential role in protecting the supply chain with end-to-end security and combating counterfeiting. With their ability to verify authenticity, report opening status and help ensure safe handling, NFC-enabled security tags can help deliver drugs to patients safely and efficiently. At the same time, for patients with chronic conditions, advanced NFC smart sensing tags can support medication adherence and remind patients to replenish their prescriptions on time. NFC tags can also deliver highly personalised and status-aware user experiences, adding value for both pharma companies and patients.
REFERENCES
- “Substandard and Falsified Medical Products”. WHO web page, Jan 2018.
- “Intellectual Property Crime Threat Assessment 2022”. EUIPO & Europol, Mar 2022.
- “Wireless Connectivity Technology Segmentation and Addressable Markets”. ABI Research, Nov 2020.
- “Global Burden of Disease (GBD)”. IHME web page, accessed May 2022.
- Sabaté S, “Adherence to Long-term Therapies: Evidence for Action”. WHO, 2003.

