To Issue 140
Citation: Jameson H, Ngan J, Shah O, “Taking the Sting Out of Vaccines”. ONdrugDelivery, Issue 140 (Nov 2022), pp 24–27.
Heather Jameson, Juliette Ngan and Omar Shah discuss the potential advantages and challenges of intranasal and inhaled vaccines in the context of global respiratory pandemics.
Cast your mind into the future and imagine the world in the grip of the next global respiratory pandemic. Once again, we are presented with the monumental task of vaccinating the world against a novel pathogen. How should we achieve this?
The current default is long lines of people stood two metres apart queuing for an injection at a clinic, masked to reduce transmission of the airborne disease, with hundreds of clinical staff and volunteers trained to administer the vaccines. Furthermore, the injectable vaccines often need cold-chain transportation if they are to reach remote communities across the world.
“Inhalation has several advantages over the traditional needle-based method, particularly when considering tackling the next respiratory epidemic or pandemic.”
Alternatively, what if it were possible to post an inhaler directly to people’s homes? Over the past few years, there has been growing interest in inhaled and intranasal delivery of vaccines. Inhalation has several advantages over the traditional needle-based method, particularly when considering tackling the next respiratory epidemic or pandemic. This article will examine the key advantages, remaining challenges and the recent progress that has been made in this field.
KEY ADVANTAGES
No Needle
The removal of the needle improves patient comfort and safety. Needles present the risk of needle-stick injuries and needle reuse, increasing the chance of cross contamination. A trained healthcare professional is required to administer the vaccine, creating a potential bottleneck for mass vaccination campaigns, particularly in non-industrialised countries and remote areas (Figure 1). Additionally, removing the needle may reduce vaccine hesitancy and increase compliance, as up to 10% of the UK population suffers from needlephobia1 and the rate in other countries may be similar.
Figure 1: A trained healthcare professional is required to administer a vaccine via injection, creating a potential bottleneck for mass vaccination campaigns.
Inhaled or intranasal delivery of vaccines could remove the need for a trained healthcare professional to be present in the majority of instances. The device could be collected from designated collection points, such as pharmacies or local grocery stores, or even delivered directly to people’s homes, for self-administration at home. However, there are hurdles to achieving this idealised situation, as discussed later in this article.
Inhaled vaccines could yield sustainability improvements too. The quantity of unrecyclable, contaminated sharps generated by covid-19 vaccine syringes in the UK alone would fill around 10 doubledecker buses (based on 150 million doses in 0.5 mL syringes). If a dry powder inhaler made from a few simple plastic parts could do the same job, it could easily be recycled into high-grade feedstock. Furthermore, dispensing with the cold chain, as discussed later, could have saved about 100 GJ of energy, equivalent to running a kettle every day and every night non-stop for two years (based on refrigeration power of 2 W per L).
Mucosal Immunity
Vaccines delivered via injection generally induce a systematic immune response, which is not specifically directed at the pathogen’s region of infection, such as mucosal sites.2 In contrast, nasal vaccines are able to instigate mucosal immunity directly at the site of infection (Figure 2),3 which may provide a higher degree of protection against respiratory pathogens.
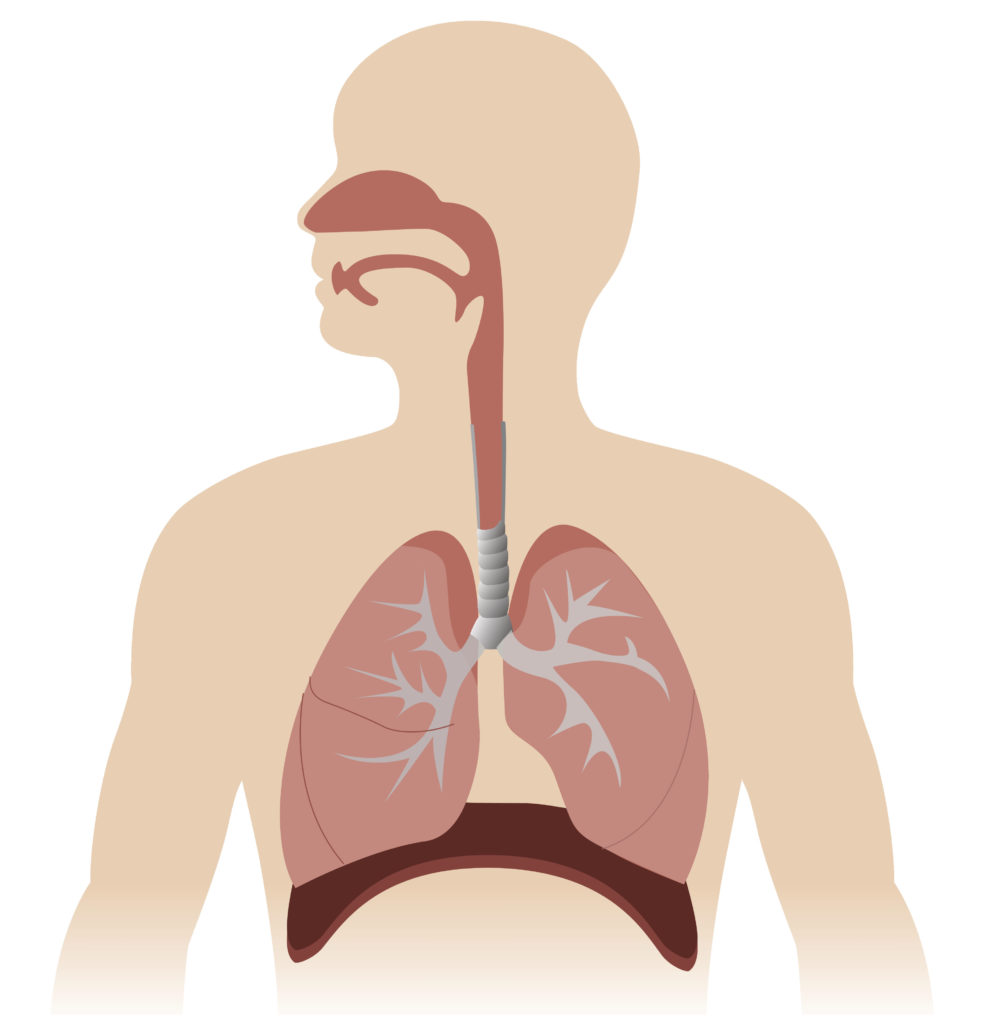
Figure 2: The respiratory system is lined by mucosal tissue. Mucosal tissue forms the physical and immunological barrier between our internal organs and the outside world.
The systemic immune response induced by intramuscular vaccines provides protection to the lower respiratory tract and prevents severe complications following a respiratory infection. However, initial infection and early disease symptoms may not be prevented because the upper respiratory tract is not protected.4 This means that the pathogens have the opportunity to invade cells, multiply and spread before they can be identified and neutralised by a systemic immune response.
“The dose accuracy will be more variable than for an injected vaccine, even if administered correctly.”
In contrast, mucosal vaccines are able to induce mucosal immunity as well as systemic immunity against a pathogen. A specialised response of mucosal associated lymphoid tissue instigated by a vaccine enables pathogens to be neutralised before they can cause an infection.2 This prevents early disease symptoms and can reduce that transmissibility of the pathogen. As has become clear during the covid- 19 pandemic, reducing transmissibility is essential in slowing down the spread of a new pathogen or strain. Moreover, this additional immune response is not limited to the site of the vaccination but also offers augmented protection in adjacent or related mucosal tissues.5 An intranasal vaccine may therefore also induce mucosal immunity in lung tissue.
Removing the Cold-Chain
Another exciting advantage of inhaled and intranasal vaccines is the option to deliver the vaccine as a dry powder, which would remove the need for cold-chain transportation and storage. Most vaccines need to be stored between 2°C and 8°C to maintain their potency. The covid-19 pandemic accelerated the progress of novel mRNA-based vaccines. These allowed for rapid development and roll-out, but with the disadvantage of extremely cold shelf-life temperature requirements. For example, the BioNTech/ Pfizer covid-19 vaccine requires storage at -80°C with a shelf-life up to six months.6
Although most can be transferred to fridge temperatures (2–8°C) for up to 30 days, the extreme cold-chain requirements of injectable vaccines can make them inaccessible for large parts of the world and can lead to vaccines being discarded, due to the time period when they can be stored above super-cooled temperatures expiring. Hence, there is interest in improving the stability of mRNA-based vaccines,6 as well as that of traditional vaccine platforms, such as by formulation as a dry powder. Formulating complex biologics into dry powders has its own challenges, as discussed later in this article, but thin-film freeze-drying is showing promising results.
The benefits of reducing the dependence on cold-chain logistics for facilitating mass vaccination campaigns across the globe cannot be underestimated – increasing access, reducing vaccine wastage and reducing the financial and environmental burden.
RECENT DEVELOPMENTS
To date, the only widely used vaccine delivered via the respiratory system is an intranasal influenza vaccine, known as FluMist in the US and as Fluenz Tetra in the UK (AstraZeneca). With the covid pandemic, there has been increased interest in intranasal and inhaled vaccines. According to Nature, around 100 nasal or oral vaccines are currently under investigation.7 Several of these have received limited approval for human use, including the CanSino Biologics (Tianjin, China) vaccine, and the Razi Vaccine and Serum Research Institute (Karaj, Iran) vaccine. Phase III trials for a number of others are underway. However, the development of the AstraZeneca vaccine as a nasal spray has hit a roadblock, with weak results in Phase I trials.8
The vaccines currently under research belong to a variety of different types, including viral vector, protein sub-unit and live virus, although they all appear to revolve around delivery in a spray or drops rather than a dry powder, which does not come with the benefits of removing the cold chain.
Active research into dry powder formulations of vaccines is less advanced than that of liquid formulations, although a 2019 study reported delivery of a dry powder influenza vaccine in ferrets with positive results.9 Some dry powder inhalers already on the market, such as TwinCaps by Hovione,10 may be suitable for the delivery of dry powder vaccines.
Despite the recent disappointing result with the AstraZeneca vaccine, the outlook for respiratory mucosal vaccination and the untapped potential of dry powder formulations is promising for the coming years.
CHALLENGES
Formulation
The respiratory system has many defence mechanisms, such as mucociliary clearance, in place to protect us from foreign micro-organisms and particles – including the constituents of vaccines. A safe, effective vaccine must be able to bypass these mechanisms to induce the appropriate immune response, without causing severe adverse effects. In order to achieve this, the selection of vaccine platform and adjuvants, if required, must be given careful consideration.
Dry powders have stability and aerodynamic advantages but formulating complex biologics into dry powders is no easy task. Physical and chemical degradation can occur during the drying process, so the choice of technique must be carefully considered. Traditional methods include spray-drying and freeze-drying (lyophilisation). However, both have disadvantages for inhaled biologics – spray-drying uses heat, which can denature proteins, and the mechanical stresses involved in freeze-drying methods can cause protein aggregation.11 Thin-film freeze-drying is an exciting new addition to the field12 that has been shown to be a viable method for converting vaccines into dry powders,13,14 giving it strong potential to become a big player.
Usability and Training
Whilst inhaled vaccines may not need a trained medical personal to be present for safety reasons, the idealised situation of self-administration at home without supervision is problematic. How do we prove that the user took the vaccine in order to authorise a “vaccine passport” or equivalent? Furthermore, how do we prove they took it correctly? Dose accuracy is inherently dependent on correct inhalation and usage by the patient for inhaled and intranasal medicines. Dry powder inhalers, for example, require a sharp intake of breath, which regular users will receive training for. A mass vaccination campaign using a dry-powder inhaler would require educating the masses effectively on the correct usage, otherwise the efficacy would be severely reduced.
Dose Accuracy
The dose accuracy will be more variable than for an injected vaccine, even if administered correctly. In addition to incorrect usage, the delivered dose could also vary due to factors such as:
- Inspiratory flow rate
- Mucosal state, such as having a cold
- Comorbidities, such as asthma
- Nasal or lung geometry
- Ambient humidity and the time taken from removing packaging to taking the drug, both of which noticeably affect dry powders due to changes in their adhesion and cohesion properties.
Furthermore, it is more difficult to measure dose accuracy for an inhaled locally acting drug.
FINAL REMARKS
Inhaled and intranasal vaccines have some key advantages over traditional needle-based methods, as highlighted in this article. However, there are challenges to overcome, if they are to be relied on for future mass vaccination campaigns.
REFERENCES
- “Injection Phobia”. Anxiety UK webpage, Accessed Oct 2022.
- Heida R, Hinrichs WLJ, Frijlink HW, “Inhaled vaccine delivery in the combat against respiratory viruses: a 2021 overview of recent developments and implications for COVID-19”. Expert Rev Vaccines, 2022, Vol 21(7), pp 957–974.
- An X et al, “Single-dose intranasal vaccination elicits systemic and mucosal immunity against SARS-CoV-2”. iScience, 2021, Vol 24(9), Article 103037.
- Amorij JP et al, “Needle-free influenza vaccination”. Lancet Infect Dis, 2010, Vol 10(10), pp 699–711.
- Kurono Y, “The mucosal immune system of the upper respiratory tract and recent progress in mucosal vaccines”. Auris Nasus Larynx, 2022, Vol 49(1), pp 1–10.
- Uddin MN, Roni MA, “Challenges of Storage and Stability of mRNA-Based COVID-19 Vaccines”. Vaccines (Basel), 2021, Vol 9(9), p 1033.
- Waltz E, “How nasal-spray vaccines could change the pandemic”. Nature, 2022, Vol 609(7926), pp 240–242.
- Madhavan M et al, “Tolerability and immunogenicity of an intranasally-administered adenovirus-vectored COVID-19 vaccine: An open-label partially-randomised ascending dose phase I trial”. EBioMedicine, 2022, Article 104298 (online ahead of print).
- Luczo JM et al, “Intranasal powder live attenuated influenza vaccine is thermostable, immunogenic, and protective against homologous challenge in ferrets”. NPJ Vaccines, 2021, Vol 6(1), p 59.
- “TwinCaps® DPI”. Hovione webpage, Accessed Oct 2022.
- Williams RO, “Improved Formulations to Enable Stable Delivery of Biologics”. Biopharm Int, Vol 35(7), pp 46–49.
- Hufnagel S et al, “The Development of Thin-film Freezing and Its Application to Improve Delivery of Biologics as Dry Powder Aerosols”. KONA Powder Part J, 2022, Vol 39, pp 176–192.
- Alzhrani RF et al, “Thin-Film Freeze-Drying Is a Viable Method to Convert Vaccines Containing Aluminum Salts from Liquid to Dry Powder”. Methods Mol Biol, 2021, Vol 2183, pp 489–498.
- Xu H et al, “Thin-film freeze-drying of a bivalent Norovirus vaccine while maintaining the potency of both antigens”. Int J Pharm, 2021, Vol 609, Article 121126.

