Citation: Sutherland G, Sherlock J-P, “The Potential of Connected Devices for Tackling Asthma”. ONdrugDelivery Magazine, Issue 76 (Jun 2016), pp 34-37.
Garth Sutherland and Jon-Paul Sherlock explain how a strategic partnership between pharmaceutical companies and digital health companies can achieve improved outcomes through joint initiatives.
“The need for a course of oral steroids, a marker of severe exacerbations, was 53% more common in children who did not use the smart inhaler compared with those who did…”
Asthma is a chronic disease characterised by recurrent attacks of breathlessness and wheezing, which vary in severity and frequency from person to person. According to the American Academy of Allergy, Asthma and Immunology an estimated 300 million people have been diagnosed and suffer from asthma globally,1 resulting in an estimated economic burden of more than US$100 billion (£78 billion).1 While asthma is the most prevalent chronic disease among children, it remains under-diagnosed and under-treated, creating a significant burden on individuals and families. And that burden is predicted to increase, with another 100 million diagnosed patients by 2025.1
Symptoms of asthma may occur anywhere between several times a day or week, and for some people become worse during physical activity or at night. Although the fundamental causes of asthma are yet to be completely understood, the strongest risk factors for developing the disease are a combination of genetic predisposition and environmental exposure to inhaled substances and particles that may provoke allergic reactions or irritate the airways. Other triggers can include cold air and physical exercise.
While asthma cannot be cured, the condition can be controlled by appropriate management, enabling patients to enjoy a good quality of life. The current gold standard treatment is a combination of short-term and long-term medications, with short-term medications used to relieve symptoms and long-term medications used to control the underlying inflammation and prevent symptoms and exacerbations.
However, the Global Initiative for Asthma (GINA) 2017 report2 showed that 70-80% of asthma patients are unable to use their inhaler correctly, meaning that these medications often do not have the intended effect (Figures 1, 2 and 3).

Figure 1: 400 million people are estimated to be diagnosed with asthma by 2025.3

Figure 2: Less than 50% adhere to their prescribed treatment.4

Figure 3: Increasing medication adherence could see a 60% reduction in hospitalisation.5
THE RISE OF DIGITAL HEALTH SOLUTIONS
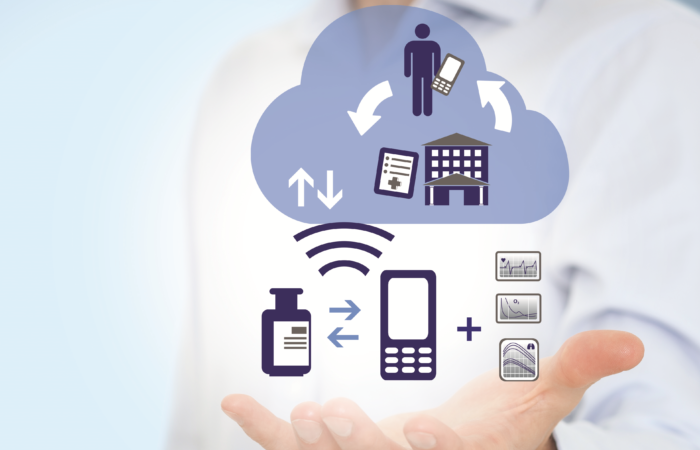
Connected devices are increasingly being adopted by consumers for health and fitness monitoring. The last few years have seen increased adoption of digital health management tools by patients, including wearables and apps, to manage their health and have access to their data. National health services around the world are seeking digital solutions to transform the treatment of chronic diseases to improve patient outcomes and ease the economic burden.
“Smart inhalers can now also monitor the inhalation technique meaning patient education to ensure that the correct inhalation technique is used can be reinforced…”
Moreover, pharmaceutical companies are partnering with digital health technology companies to accelerate research and development of devices and platforms to bring these digital tools to patients. Enabling self-monitoring and management of long-term conditions has been identified as a way for healthcare systems to reduce the overall cost of care.
Healthcare providers and pharmaceutical companies are increasingly using technologies to engage patients with chronic conditions in directed self-management of their conditions. Empowering patients to engage with their condition, through supported self-management and personalised approaches, can instil an increased sense of responsibility. This, in turn, can result in appointments with clinicians becoming less frequent.
Once deployed, digital platforms that link a monitoring or management device to cloud-based software for storing data, can be accessed by both the patient and the healthcare practitioner. This helps to ensure that exacerbations in a patient’s condition can be identified and action can be taken to reduce emergency admissions and, in extreme cases, avoid fatal complications.
INCREASING ADHERENCE WITH SMART INHALERS
One of the greatest barriers to improving the health of asthma patients is drug adherence. Various tactics have been tried to increase adherence, from professional education and advice from clinicians and pharmacists to family support. Unfortunately, all have proved to be complex, time-consuming and costly, with modest results.
Smart inhalers have been recognised as a novel approach to promoting adherence by monitoring medication use for respiratory diseases, including asthma.
Smart inhalers can be used by adults and children, with initial research showing a willingness among the asthma population to carry a connected device. This means there is a significant opportunity for the use of smart inhalers so that people with asthma, healthcare professionals and national health services can use the data to help improve outcomes and reduce costs.
With all digital solutions, some training will be needed to familiarise patients and clinicians with the device and associated software. This may include fitting the device correctly and checking inhaler technique. Patients will also need to be advised of what data are captured and how they are able to view them. Smart inhalers can now also monitor the inhalation technique meaning patient education to ensure that the correct inhalation technique is used can be reinforced.
DIGITAL HEALTH INITIATIVES
An example of the digital health initiatives taking place between pharmaceutical companies and digital health companies is the strategic partnership between Adherium and AstraZeneca. The shared aim is to support respiratory patients through the introduction of new technologies and innovations in digital healthcare.
This digital health initiative is focused on demonstrating that new technologies can be combined with medication to help patients achieve improved outcomes. Because these technologies evolve incrementally in a real-world setting with the experience and input from healthcare professionals and patients, they are not only making great strides towards delivering the future of digital health, but most importantly, adding value to existing treatment options.
Turbu+
Turbu+ is the first programme to come out of this initiative, with the aim of reinforcing the efficacy of AstraZeneca’s existing inhalers in real life by establishing the right behaviour early in a new treatment. This will be achieved through provision of reminders and motivational messages to patients by incorporating Adherium’s Smartinhaler™ technology, which has been shown to improve adherence when compared with standard care.
The enhanced Smartinhaler™ device sends the captured data via Bluetooth to an app, and the device programme tracks the patient’s treatment regimen. As well as supporting patients, the device provides their healthcare professionals with secure and accurate information, which gives them a deeper understanding of how their patients are using their medication in real life. It not only equips them with information on actual medication use to make better treatment decisions and treat the patient efficiently during consultation, but reassures them to what extent their treatment plan is being followed.
GROWING CLINICAL EVIDENCE OF IMPROVED OUTCOMES
Patient adherence to prescribed medications represents a significant issue for chronic disease management. Approximately 50% of adults and children on long-term therapy for asthma fail to take medications as directed at least part of the time.6 In the UK a child is admitted to hospital every 20 minutes because of an asthma attack.
Numerous clinical studies and peer-reviewed papers highlight the need for a widespread digital solution, such as smart inhalers, that promotes adherence to medication. Meanwhile, clinical studies have shown that asthma patients are achieving 80% adherence to preventative medication when using a smart inhaler device with a 61% reduction in oral steroid use.
“The big data that can be gathered from devices and platforms will determine the way in which digital solutions can be improved and, in turn, how care can be improved…”
For example, the UK-based year-long STAAR study, carried out at Sheffield Children’s Hospital and led by the University of Sheffield’s Dr Robert Morton and colleagues, aimed to assess whether introducing digital adherence monitoring into routine practice could improve clinical outcomes in children with poorly-controlled asthma.7
In the study 38 children were in the intervention group using adherence monitoring smart inhaler with medication reminders and feedback in the clinic, and 39 children received usual care as part of a control group. Drug use data were collected and children’s health outcomes were assessed at each three-month follow-up. The study found that adherence to prescribed medication averaged 70% in children using a smart inhaler compared to 49% in the control group (p<0.001).
The study also found that increased medication adherence through use of the smart inhaler device and data platform was maintained over the 12-month period of the study, with nearly half of the children using the smart inhaler maintaining average adherence rates of >80% over the 12 months.
The study also found improvements in outcomes. The adherence improvement among children using the smart inhaler was associated with significant reduction in asthma exacerbations – episodes of progressively worsening shortness of breath, coughing, wheezing and chest tightness – which can be life threatening. The need for a course of oral steroids, a marker of severe exacerbations, was 53% more common in children who did not use the smart inhaler compared to those who did. (p=0.008).
Furthermore, the hospitalisation rate was five times greater in the control compared with the intervention group (p<0.001). This approximates to the prevention of 12 hospitalisations in one year among the children in the intervention group, making a cost-saving argument for introducing smart inhalers into routine practice.
Through the course of the study, the clinical benefits observed within the intervention group increased compared with the usual care group, particularly at nine and 12 months, with the intervention group requiring fewer courses of oral steroids, hospital admissions, days off school and GP/emergency department visits.
The study concluded that existing digital technologies should be introduced to patients to improve asthma care and that more should be done to ensure that people with asthma are able to benefit from a connected way of managing their condition (Figure 4).

Figure 4: The intervention group saw an increase in adherence to medication and a reduced need for courses of steroid medication.7
CONCLUSION
Multiple clinical studies have already demonstrated that smart inhalers can lead to improved adherence and, even more encouragingly, associated benefits such as reduced hospital admissions. The introduction of smart inhalers therefore has the potential to realise savings for healthcare providers, while also enabling clinicians to gain increased insights into their patients’ behaviours and intervene for improved health outcomes.
The big data that can be gathered from devices and platforms will determine the way in which digital solutions can be improved and, in turn, how care can be improved. While efforts to incorporate digital solutions into national health services will take time and upfront costs, the benefits will outweigh the initial integration with improved patient health seeing reduced healthcare costs.
REFERENCES
- “Asthma Statistics”. AAAAI Web Page, 2017. (www.aaaai.org/about-aaaai/newsroom/asthma-statistics)
- Global Initiative for Asthma Web Page, 2017. (www.ginasthma.org)
- Anandan et al, “Is the prevalence of asthma declining? Systematic review of epidemiological studies”. Allergy, 2010, Vol 65(2), pp 152-167.
- Horne R, “Compliance, adherence, and concordance: implications for asthma treatment”. CHEST, 2006, Vol 130(1_suppl), 65S-72S.
- Williams et al, “Relationship between adherence to inhaled corticosteroids and poor outcomes among adults with asthma”. J Allergy Clin Immunol, 2004, Vol 114(6), pp 1288-1293.
- Boulet L-P, Vervloet D, Magar Y, Foster JM, “Adherence: the goal to control asthma”. Clin Chest Med, 2012, Vol 33, pp 405-17.
- Morton RW et al, “STAAR: a randomised controlled trial of electronic adherence monitoring with reminder alarms and feedback to improve clinical outcomes for children with asthma”. Thorax, 2016, Vol 72, pp 347-354.