To Issue 183
Citation: Draudt G, “Intravitreal Injections – What is Causing Patients to Discontinue Therapy and Why is it so Hard to Fix?”, ONdrugDelivery, Issue 183 (Mar 2026), pp 20–23.
Gregg Draudt considers the significant issue of treatment discontinuation in intravitreal therapies, digging into the root causes of poor adherence and what will be required to reverse this trend in a way that caters to the full range of stakeholders, who often have contradictory requirements.
If you have attended a recent ophthalmology conference or looked at the academic literature, you will have heard that the issues of patient burden and non-adherence associated with intravitreal injection therapy (IVT) are currently a major point of concern. The estimate is that as many as 50% of patients stop their therapy within four years. Investigating the influential factors that cause people to discontinue treatment, this article provides a system-level view of IVT, highlighting how misaligned stakeholder priorities can make things harder for patients.
HOW DID WE GET HERE?
In the mid-2000s, the landmark approvals of anti-vascular endothelial growth factor (anti-VEGF) drugs – especially Lucentis (ranibizumab, Genentech) – revolutionised treatment for neovascular diseases such as wet age-related macular degeneration, diabetic macular oedema and retinal vein occlusion. Over the following 15 years, anti-VEGF use exploded to millions of injections per year.
While being hailed as an amazing therapy, anti-VEGF patients require an anxiety-laden monthly dosing regimen, leading to an accumulation of negative treatment burdens on the patient. It also transformed clinical practice for retinal ophthalmologists, creating entirely new business models where multiple days a week are spent doing long strings of IVT, delivering as many as 60 or more injections per day per ophthalmologist (in the US).
In the 2020s, there has been an increasing focus on innovation and optimisation to begin to address the growing number of patients missing appointments and abandoning IVT regimens entirely. These innovations have focused on reducing injection frequency via different formulations and durations, improving clinical workflows and other ways of supporting long-term adherence.
Among the accumulated stack of issues pushing patients away from IVT, the heightened emotional anxiety felt around the injection itself has the strongest negative impact. These highly emotional aspects can be made far less intimidating through a combination of improved technology, human factors considerations and product design. However, the pathways to these patient-centric drug delivery improvements present substantial structural and business model hurdles.
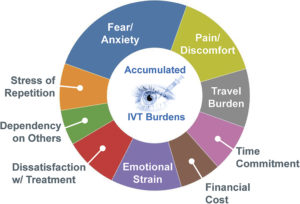
Figure 1: Accumulated burdens (side-effects play a significant role in these burden groupings).
WHAT ARE THE PATIENT BURDENS?
From the patient’s perspective, IVT is defined by two dominant challenges – the time it takes and the anxiety it produces. However, a look at the complex interplay of factors that impact patient experience shows that there is no simple “silver bullet” solution (Figure 1 & Table 1). Addressing the discontinuation of treatment will therefore require significant systemic changes both to the patient journey and clinical workflow. Some of the factors causing patients to discontinue therapy and which add to the levels of patient fear and anxiety include:
- Process Inconsistency: There is no one standard method or process for implementing IVT – changes in process trigger heightened emotions
- Needle in the Eye: The experience of the bare needle is frightening
- Speculums: Most clinics use speculums for keeping the eye open, exacerbating the sense of helplessness and impending danger for patients
- Fixation: Patients’ own involvement in holding their eye steady during the injection process is frightening – they feel responsible for possible serious injury to the eye
- Subconjunctival Haemorrhaging: “Red Eye Roulette” happens in an estimated 10% of injections, can last for weeks and can cause embarrassment and social isolation until it resolves
- Increased Intraocular Pressure (IOP): Heavy eye and eye discomfort are the short-term issues of increased IOP, and repetitive or prolonged pressure on the optic nerve can cause more serious long-term visual degradation
- Multiple Steps: IVT delivery requires many steps, including antiseptic and anaesthetic, which prolongs the process, offers additional discomfort and is often messy
- Side-Effects: Patients often fear critical side-effects, such as endophthalmitis, as well as other non-critical ones
- Logistics: Other significant burdens less tethered to the drug delivery process itself include travel time, the need for caregiver support, injection frequency and cost.
| Patient Experience | Cause |
| Pain or irritation | • Chemical irritation • Mechanical irritation • Spike in IOP |
| Visual disturbance | • Vitreous opacities (floaters) • Turbulence and air bubbles |
| Bloodshot eyes | • Burst blood vessel |
| Affected vision or blindness | • Endophthalmitis (infection) • Traumatic cataract • Retinal detachment |
Table 1: Potential side-effects.
HAVE RECENT INNOVATIONS HELPED?
The primary focus of innovation has been reducing the frequency of injections, and there has been a proliferation of innovative pharmacological variations and drug delivery systems that champion the value of greater durability. Durability, in this context, refers to how long the therapy remains active in patients after a dose is delivered. Logically, when a dose of therapy lasts longer, patients will require fewer, less frequent trips to the clinic – this should result in a reduced patient burden and significantly fewer therapy discontinuations over time. Some remarkable devices have been and continue to be developed to help deliver more durable solutions, including port delivery, implants and gel-centric technologies.
“ONE STUDY FOUND THAT SOME GROUPS OF PATIENTS WHO WERE MOVED TO 12-WEEK OR LONGER INTERVALS WERE MORE LIKELY TO STOP TREATMENT THAN THOSE WHO CONTINUED ON SHORTER SCHEDULES.”
To understand the impact of these remarkable innovations, studies have aimed at assessing the association between treatment interval and the likelihood of patients discontinuing anti-VEGF treatment. What these studies have revealed is surprising. Real-world studies (including Bakri et al, 20221) looking at injection schedules and long-term treatment behaviours have shown that less frequent injections do not automatically lead to better patient follow-through. In fact, one study found that some groups of patients who were moved to 12-week or longer intervals were more likely to stop treatment than those who continued on shorter schedules.
While further research is needed, received wisdom suggests that when patients come in less often, they may feel that their disease is stable or may simply lose the routine of ongoing care. Therefore, even though longer-lasting treatments help reduce the burden of clinic visits, it is possible that greater durability alone is not enough to keep patients adherent. Other permutations of durability might offer better results. If, for instance, there were just a single one-and-done injection – as promised by some gene therapies – that would certainly be a winner. Short of that, effective care will require addressing the burdens more holistically.
“AN IVT DEVICE SOLUTION THAT TRULY ACCOMMODATES THE SYSTEM-LEVEL REQUIREMENTS IS NOT LIKELY TO BE A CONVENTIONAL ‘BETTER NEEDLE’ OR INCREMENTAL IMPROVEMENT OF THE INJECTION DEVICE.”
WHAT DOES HOLISTIC MEAN IN THIS CONTEXT?
A more patient-centric IVT delivery device and associated workflow will only be seen as a great innovation when it elegantly integrates into the wider set of success factors. Furthermore, lowering the patient burden may or may not be enough to substantially lower the discontinuation rate, as the patient burden is just one of the critical puzzle pieces in the overall IVT stakeholder ecosystem. The next generation of IVT drug devices must fit into a broader and dynamic ecosystem shift while also making non-adherence a much less common outcome. An IVT device solution that truly accommodates the system-level requirements is not likely to be a conventional “better needle” or incremental improvement of the injection device – it will have to function as a system-enabling device that simultaneously changes clinical confidence, workflow economics, patient burden and reimbursement logic.
WHO ARE THE STAKEHOLDERS IN THE IVT ECOSYSTEM?
There are quite a few stakeholders involved in a holistic IVT solution – some wielding more clout than others. The primary stakeholders are:
- Patients
- Clinics and practices
- Private equity
- Value-based health systems
- Insurers and payers
- Pharma companies
- Device companies
- Ophthalmologists.
A device design that benefits all IVT stakeholders must overcome a number of divergent – and sometimes contradictory – goals (Figure 2). Some of these include reducing patient burden, preserving clinical flexibility, maintaining clinic economics, protecting pharma value, satisfying payer cost logic and scale within existing health system workflows. Understandably, each set of stakeholders has different priorities, and these do not all point to the same solutions. A device that truly “works” for IVT must resolve most, if not all, of these structural conflicts.
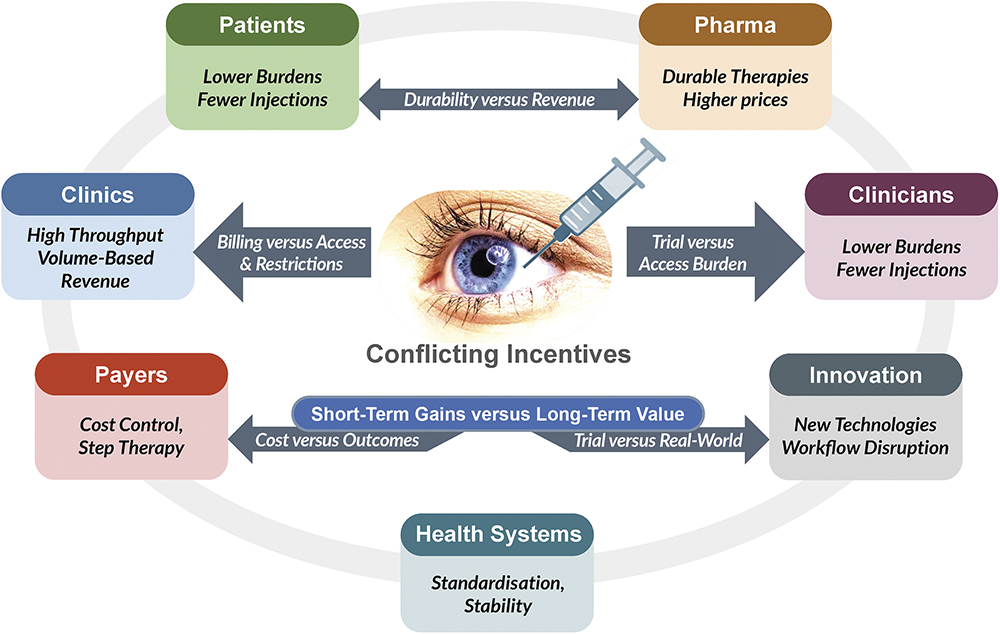
Figure 2: Inherent conflicts within the IVT ecosystem.
When looking at the IVT delivery challenge from the separate stakeholders’ perspectives, solutions could quickly lead in different directions:
- Patients: Want to reduce their lived burdens by lowering anxiety without increasing risk. Patients value reliability, predictability and the user experience as much as having fewer injections.
- Clinicians: Want to preserve clinical control and flexibility. Clinicians are very reluctant to change their process.
- Practices: Want to protect economic sustainability while improving clinic throughput. Clinics will adopt burden-reducing solutions only if the economics remain neutral or positive.
- Pharma Companies: Want to protect value while enabling differentiation. Pharma companies will support devices that reframe durability as value, not lost volume.
- Payers: Want to reduce the cost of care. Payers value predictable, measurable evidence, not novelty.
- Healthcare Systems: Want to standardise procedures, minimise training and reduce bottlenecks. Healthcare systems care about maintaining stability while improving care delivery.
- Device Innovators: Want to quietly resolve conflicts between other stakeholders. For device developers, economic benefits drive adoption, but patient safety, enhanced workflows and more ideal usability motivate change.
WHERE TO START?
Mapping and keeping track of all these variables (Figure 3) is very important – but knowing when each stakeholder group should be considered is fundamental to finding a way forward. One common set of factors that all stakeholders must support is that the therapy is safe, effective and not alienating for patients. Therefore, the starting point for finding better solutions begins with developing a deep, holistic understanding of each of the pain-points, burdens and side-effects.
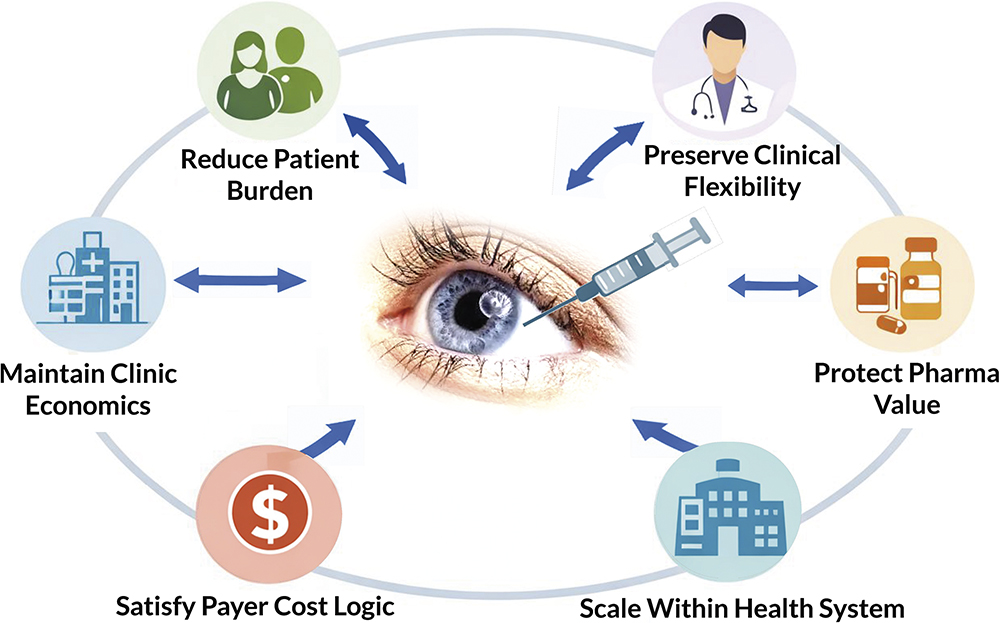
Figure 3: Future IVT devices must simultaneously benefit multiple stakeholders with conflicting priorities.
To get there, design teams must attempt to prioritise the user experiences that are often muddled together under headings such as fear, anxiety and pain, after which they can begin developing potential solutions to eliminate or minimise enough of these negative, alienating aspects to have a profound impact on the patient experience. Once a carefully designed solution – one that will keep a significant percentage of otherwise disenfranchised patients engaged in treatment – has been prototyped and tested with patients and a set of key initial stakeholders, it will then need to be tuned and refined to accommodate the needs of the remaining stakeholders within the IVT ecosystem.
REFERENCE
- Bakri SJ et al, “Anti-Vascular Endothelial Growth Factor Treatment Discontinuation and Interval in Neovascular Age-Related Macular Degeneration in the United States”. Am J Ophthalmol, 2022, Vol 242, pp 189–196.

