Citation: Volmer J, Lenn J, Brown M, “Assessing the Nasal Drug Delivery Landscape”. ONdrugDelivery, Issue 119 (Apr-May 2021), pp 37–40.
Jon Volmer, Jon Lenn and Professor Marc Brown discuss the role of reconstituted nasal epithelium in intranasal drug delivery.
Intranasal delivery has always been a tantalising target for topical and systemic drug delivery due to readily available access, high levels of vascularisation, a large surface area and the ability to deliver directly to the central nervous system (CNS). As one of the primary sites of infection for respiratory diseases, it is also a logical target for vaccine delivery. Delivery is relatively painless, onset of drug action can be rapid and formulations can be easily administered in emergency situations – for example, the Narcan (naloxone) intranasal spray (Emergent BioSolutions, Gaithersburg, MD, US).
As with all methods of drug delivery, development of new intranasal formulations and treatments can only advance as fast as the existing models allow. Therefore, it is always important to reassess and improve upon the models that are currently available. MedPharm is always looking for ways to use its expertise to improve the formulations and the models used to develop these potential products. Reconstituted nasal epithelium (RNE) is one of the latest advancements MedPharm is using to help clients de-risk and expedite intranasal delivery, thereby giving a strategic competitive edge.
NASAL DRUG DELIVERY
The epithelial surfaces of the nose can be divided into four general regions. The most distal of these is the nasal vestibule, characterised by a squamous mucosa that slowly transitions to keratinised skin towards the nares. Just proximal to the nares is the nasal cavity, the largest of the four regions. It is macroscopically characterised by three rigid shell-like protrusions known as turbinates. These structures serve to filter, warm and humidify incoming air. The convoluted surface of the nasal cavity slows incoming air, promoting the deposition of particulate matter on the nasal mucosa. A high level of vascularisation in these structures provides the moisture and warmth required to condition incoming air for optimal gas exchange. Combined, these four regions make an excellent target for drug delivery.
Droplets from sprays or particulate from powders will enter the nasal cavity, encounter the turbinates and adhere to the mucosal surface, where they gain access to the vasculature. Delivery is complicated, however, by the presence of tight junctions, active ciliated epithelial cells and a protective layer of mucus on the epithelium. Particulates are trapped in the mucus layer, which is swept along by the cilia to the oropharynx, where they can be cleared by swallowing, coughing or expectoration. This rapid clearance necessitates similarly rapid release and absorbance before the drug product is removed from the target epithelium.
Situated above the nasal cavity is the olfactory mucosa, a specialised region of the nose characterised by the presence of olfactory neurons and supporting sustentacular cells. The olfactory mucosa provides another dimension to nasal drug delivery by providing direct access to the CNS, bypassing the blood-brain barrier and making it possible to effectively deliver a drug to a CNS target within seconds. The mechanism by which this happens is still the subject of some discussion, but likely involves transport of the drug through the paracellular space between olfactory axons or via the conduit-like lamina propria in which olfactory axons are bundled.1
RECONSTITUTED NASAL EPITHELIUM
Historically, one of the more common in vitro models used to screen various formulations for nasal delivery is excised animal nasal mucosa (e.g. sheep) mounted in a Franz cell. Formulations are applied to the apical side of the tissue, and receptor solution is sampled from the basolateral side to determine formulation performance. This model has proven quite successful in the development of nasal drug formulations for decades and MedPharm has used it with many clients in the development of several drug products.
However, like all models, this model of nasal drug performance has some limitations for a more complete picture of nasal drug delivery. Although passive barriers such as basement membrane and matrix binding remain intact, the active components such as tight junctions, mucus production, cilia and other cellular activities do not. To build on and improve this model, MedPharma has developed an RNE model. This model system uses primary human nasal epithelial cells, harvested from the mucosa at the turbinates, regrown on permeable inserts and stimulated to develop into a well differentiated nasal epithelium.
Differentiation of airway epithelium in air-liquid interface (ALI) cultures has been described since the early 1980s.2 These models have historically been highly complex and difficult to reproduce, and reliable sources of primary cells are hard to obtain. MedPharm has leveraged the recent technological advancements, years of tissue culture and organoculture expertise to build on and develop new methods that allow for a reproducible RNE construct that can be used in a high throughput fashion to develop and optimise intranasal formulations.
Nasal epithelial cells are harvested from a donor using a nasal swab and resuspended in a gentle proteolytic media bath. Epithelial cells are isolated from resident immune cells and fibroblasts and then expanded in monolayer culture using specific epithelial growth media. Cells are then seeded onto porous membranes, supported over a bath of similar growth media. Over the course of about a week, cells expand and become fully confluent, beginning the differentiation process. At this point, MedPharm’s proprietary media is changed and the apical surface is kept dry to promote full differentiation, which occurs over the span of 3–4 additional weeks. Over this time, the epithelial cells adopt a pseudostratified columnar compound epithelial layer. These cells exhibit mucus production, ciliary activity and tight junctions, and this architecture typically persists for approximately four weeks.
Development of barrier function in RNE cultures is confirmed by measuring transepithelial resistance (TEER). Tight junctions form an electrochemical barrier between the basolateral and apical chambers, and this can be measured by determining electrical resistance. This slowly builds over time to a stable state after about three weeks and is dependent on the presence of differentiation media (Figure 1). TEER can be measured immediately prior to administration of formulation, to ensure that only intact and well-differentiated constructs are used in analysis.
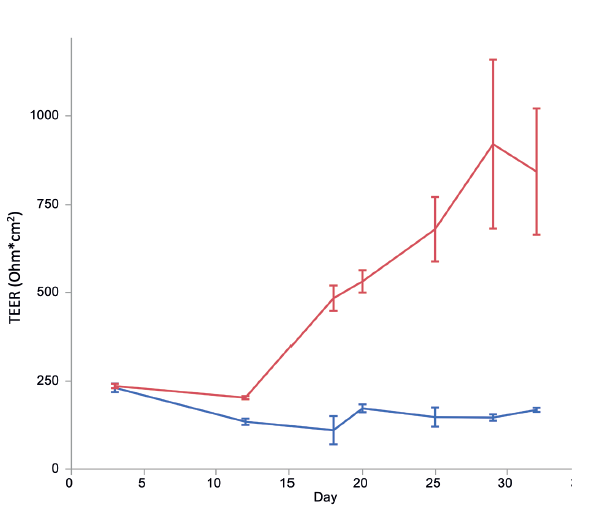
Figure 1: Primary human nasal epithelial cells were seeded on a permeable membrane which was suspended in media. The apical side of the membrane was also covered in media. Cells were allowed to grow until confluent (approximately 10 days), at which point they were switched to air-liquid interface and given differentiation media, or left in the growth conditions. TEER (Ohm*cm2) is plotted versus time (days) for cells treated with growth media (blue) or differentiation media (red). Error bars are SEM, n=5.
Oxycodone and buprenorphine have been the subject of clinical studies in which intranasal administration has been compared with intravenous administration.3 In an effort to determine if the same qualitative trends would translate from those studies to the MedPharm model, buprenorphine and oxycodone formulations were prepared to closely mimic the formulations used in the clinical studies. In the trials, the oxycodone formulation was reported to have approximately seven-fold higher Cmax than the buprenorphine formulation. The overall ranking was similar, with some slight differences in the magnitude of differentiation (Figure 2).
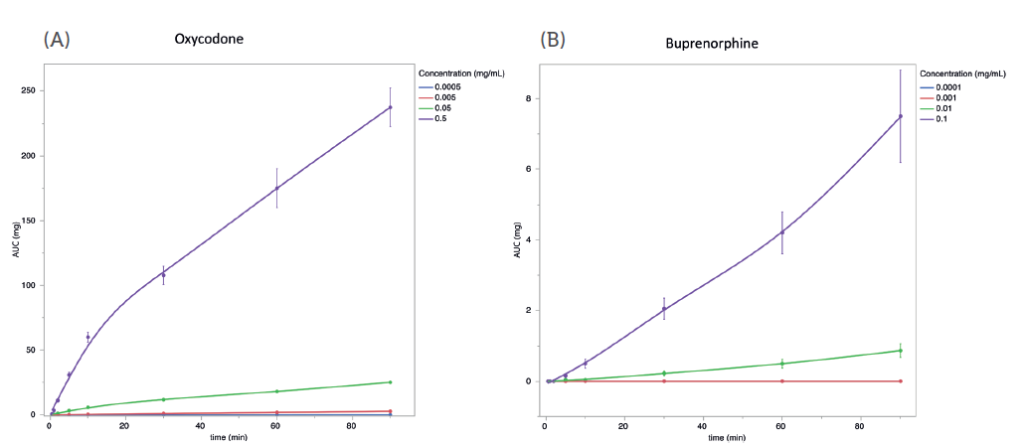
Figure 2: 10 μL of oxycodone (A) or buprenorphine (B) in saline solution at the indicated concentrations was applied to the apical side of well-differentiated RNE. At the indicated times, basolateral media was collected, and replaced with an equal volume of fresh, prewarmed media. Total permeated quantity at each timepoint (AUC) was calculated and plotted versus time. Error bars are SEM, n=5.
To further determine formulation discrimination in the RNE model, a dose-response series was conducted for the oxycodone formulation. Four concentrations of oxycodone formulation were prepared and administered apically to RNE constructs (n=5). At designated intervals, a sample of media from the basolateral side was collected, and the basolateral volume replenished. A total cumulative amount was calculated for each time point. The total cumulative amount at all time points past two minutes showed a linear response to formulation concentration (R2 >0.9) showing a dose-response for the oxycodone formulations (Table 1).
| Time (min) | R2 |
| 0.5 | 0.85 |
| 1 | 0.87 |
| 2 | 0.92 |
| 5 | 0.95 |
| 10 | 0.95 |
| 20 | 0.95 |
| 30 | 0.95 |
| 60 | 0.94 |
| 90 | 0.96 |
Table 1: Formulations of oxycodone were prepared at four concentrations. Total cumulative amount was calculated for each formulation at each of nine time points. Total cumulative amount for each time point was plotted against formulation concentration to determine if there is a linear relationship between total cumulative amount and concentration of API in formulation. For each time point after two minutes post-administration, a strong relationship (R2 >0.9) was observed.
CONCLUSIONS AND FUTURE DIRECTIONS
RNE opens new avenues of investigation in drug delivery to or via the nasal route. In addition to being a closer approximation of the nasal mucosal barrier than traditional animal tissue models, RNE cultures allow for additional analyses that are not possible using ex vivo tissue. Mucosal epithelium contains influx/efflux pumps, which can transport drugs into or out of the cell layer. A host of constitutive and inducible metabolic enzymes can alter drugs as they pass through the cell layer. Active changes in the type and quantity of mucus in response to formulation delivery can impact permeation. Certain excipients can have active or indirect effects on tight junctions. This model can also be used to monitor direct effects of drugs targeting the nasal epithelium due to cellular responses and can be used to assess local toxicity or irritation.
This model has been extended to include nasal infection. The advent of the covid-19 pandemic has highlighted the significance of respiratory disease, the importance of good models of infection and pathology, and the role of the nasal epithelium in infection and viral propagation. MedPharm has begun the development of models of coronavirus infection in the nasal epithelium, using reconstituted nasal epithelium. Modelling infection in RNE instead of cell lines (such as HBE4 and A549) bypasses many of the well-known shortcomings associated with cell lines in general. Well-differentiated primary cells more closely mimic in vitro physiology, cell signalling and architecture. This has special importance in viral infection models, as the conditions in the host cell (such as protease and receptor expression) can generate artificial selection during propagation and infection. This selection can cast doubt on results from such work. Although the use of RNE does not eliminate this effect, it can reduce it.
The RNE model can also be easily adapted for bronchial epithelium, allowing for the testing of a host of new formulation types, such as those for the treatment of asthma, COPD, bronchiectasis and cystic fibrosis. In the case of cystic fibrosis (and other genetic diseases), epithelial constructs can be grown from cells collected from the affected patient population during routine medical care.
RNE and other airway epithelial models can further expand their utility through the use of co-culture systems. Co-culture with inflammatory cells can allow for invasion assays, comparing the performance of anti-inflammatory and antibiotic drugs. Reconstructed airway tissue can be co-cultured with airway smooth muscle to compare efficacy of drugs treating airway hyper-responsiveness in asthma. Due to the long-term viability of these constructs, remodelling effects from irritants such as smoke, diesel exhaust particles and other environmental particulates can be monitored, and recovery from insult compared.
It is always the goal to develop the next more useful model, and thereby advance drug discovery. With this RNE model, MedPharm has increased the utility of available models for intranasal delivery.
REFERENCES
- Erdo F et al, “Evaluation of intranasal delivery route of drug administration for brain targeting”. Brain Res Bull, 2018, Vol 143, pp 155–170.
- Lechner JF et al, “Clonal growth of normal adult human bronchial epithelial cells in a serum-free medium”. In Vitro, 1982, Vol 18(7), pp 633–642.
- Dale O, Hjortkjær R, Kharasch ED, “Nasal administration of opioids for pain management in adults”. Acta Anaesthesiol Scand, 2002, Vol 46(7), pp 759–770.
Previous article
CONSIDERATIONS FOR OPHTHALMIC DRUG DELIVERY AND DEVELOPMENTNext article
FORMULATION OPTIONS IN NASAL DRUG DELIVERY
