To Issue 155
Citation: Cuevas Brun E, Hsu Y-M, “Customisable Nebuliser Platform to Monitor Adherence”. ONdrugDelivery, Issue 155 (Dec 2023), pp 16–20.
Edgar Hernan Cuevas Brun and Yuan-Ming Hsu discuss the value that customisable nebuliser platforms bring to patient adherence.
“Deterioration of a patient’s health condition is the primary consequence of poor treatment adherence.”
Innovative approaches in inhaled drug delivery systems have contributed to improvements in delivery performance and overall user needs. These approaches aim to address some longstanding shortcomings, which have remained unsolved for several decades. One such shortcoming has been widely described as the challenge of accurate adherence monitoring. Adherence monitoring plays a crucial role in understanding how effectively patients follow their prescribed treatments and how that relates to the subsequent outcomes.
Deterioration of a patient’s health is the primary consequence of poor treatment adherence. Simultaneously, the lack of effective adherence to chronic disease management is often associated with an extensive economic burden on healthcare systems. Patients may experience more severe symptoms when not following the treatment prescribed to them by health professionals. In many cases, these scenarios can lead to more frequent emergency hospital visits, further increasing the financial stress on healthcare systems.
Among the respiratory diseases that are often treated by the use of inhaled devices, asthma in particular is associated with recurrent hospital visits due to a higher number of exacerbations.1,2 Inconsistent intake of medications can also impact the treatment of other conditions, such as chronic obstructive pulmonary disease, cystic fibrosis and pulmonary arterial hypertension.
Over the past few years, the incorporation of adherence monitoring solutions has gradually been implemented in new inhalers and nebuliser systems in increasing numbers, either as add-ons or integrated forms. A large number of these solutions rely on Bluetooth connectivity for this purpose. The goal has been to provide patients, healthcare professionals and healthcare systems with a tool that can alleviate some of the burdens caused by lower adherence levels.
The focus is on monitoring approaches that could eventually result in higher adherence levels. Although, initially, studies did not show significant improvements in adherence or were inconclusive to some extent, more recent studies are beginning to shed light on this matter. This has ignited a sense of contribution to the development of connected devices, cementing a space for their future adoption, thanks to constant advances in the internet of things (IoT).3,4
A NEBULISER SOLUTION, ADHERESP = ADHERENCE + RESPIRATORY
Pharmaceutical companies developing new inhaled therapies are showing increasing interest in incorporating more advanced nebulising systems. These include mesh nebulisers, which are characterised by their portability and high delivery performance, especially when used with specific medications as drug-nebuliser combination products. With these effective solutions, attention is also being paid to to the inclusion of higher-end products that not only focus on performance efficiency, but also aim to integrate new features. Connectivity is at the top of this list of features.
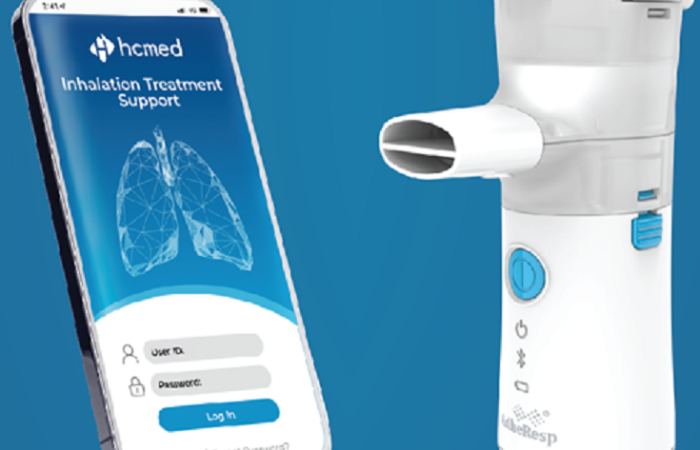
Driven by the demand for adherence monitoring and the potential of connectivity to fulfil this requirement, the smart, breath-actuated mesh nebuliser, AdheResp, was designed to record data that could effectively support these needs (Figure 1). The AdheResp nebuliser was developed with the latest Bluetooth technology to transmit data from the handheld device to a mobile application, which can, in turn, be linked to a medical cloud service platform. The primary goal is to support adherence monitoring and provide pharmaceutical partners with the option of including connected features in their new combination products, while also benefiting from other functions, including breath-actuation and activation.

Figure 1: AdheResp – a smart, breath-actuated mesh nebuliser.
“Driven by the demand for adherence monitoring and the potential of connectivity to fulfil this requirement, the smart, breath-actuated mesh nebuliser, AdheResp, was designed to record data that could effectively support these needs.”
The standard version of the AdheResp nebuliser can transmit three major sets of data to a mobile device. These data sets include time information, battery status and pressure changes recorded as part of the breath-actuation. These data sets were selected based on their identification as key parameters for supporting adherence monitoring and usability during extensive product research. Additionally, supported by the customisable nature of the AdheResp platform, the optimised versions of the AdheResp nebuliser offer expanded possibilities for connectivity beyond these aspects. This includes other usability and traceability options, such as inhalation feedback, guidance on successful inhalation and tracking of medication batches.
As new demands in adherence monitoring are identified, it can be expected that newer functions could be made available thanks to the close collaboration between software or mobile app developers, medical cloud service providers and smart mesh nebulisers, such as HCmed Innovations’s customisable AdheResp platform.
STANDARD AND CUSTOMISABLE FEATURES OF THE ADHERESP NEBULISER PLATFORM
During the development process of the AdheResp nebuliser, some key functions were identified to monitor adherence, with the aim of meeting the needs of the different stakeholders involved in a patient’s treatment. Pharmaceutical companies, healthcare professionals and patients themselves are part of this intertwined system, each with unique needs but all sharing a common goal – supporting adherence for the benefit of patients.
While batch traceability and tracking treatment efficiency may be more valuable for pharmaceutical companies, patients would perceive greater benefits from real-time data transmission and data recording, which is also indispensable for healthcare professionals. Some of the existing and customisable functions that could be captured within the AdheResp platform (in some cases in combination with a customisable mobile app and medical cloud service) are discussed here.
Treatment Data Recording
Data recording is an essential tool that benefits all parties during adherence monitoring. The AdheResp nebuliser can transmit data, including the time of treatment, length of individual treatment period, changes in pressure during treatment (associated with the breathing profile) and feedback on the expected inhalation period, which can be interpreted as a successful/unsuccessful inhalation. These records can be shared between patients, healthcare professionals, healthcare systems and even pharmaceutical companies via the incorporation of medical cloud services that enable the uploading of the transmitted data to a mobile app. Furthermore, and also supported by a mobile app, recorded data could be exported to files, which could then display selected data based on specific dates or timeframes.
“To support adherence in younger users, the implementation of games in a mobile app could serve as the means to engage with the use of the nebuliser.”
Drug Traceability
This tool is associated with the transmission of batch information from a vial or ampoule at the time of treatment. The AdheResp nebuliser features an activation mechanism that uses near-field technology (NFC) to activate the nebuliser before each treatment. It is possible to integrate the NFC tag into the label of a vial or ampoule to activate the nebuliser and, at the same time, transmit the information of the nebulised drug. The information, which is initially stored in the device, can then be transmitted to the mobile app for data recording. Drug traceability is another major option that benefits all parties in monitoring patient adherence to the prescribed treatment, enabling follow-up during subsequent appointments.
Tuneable Delivery Platform
For certain indications, a specific triggering span during inhalation or output rate may be required. To address this need, the AdheResp nebuliser can be activated using different NFC tags, which contain customised algorithms to enable the appropriate operation during activation. This activation configuration can be transmitted to the mobile app, providing further information for periodic review within the treatment scope, as well as space for adjustments during the overall treatment. This function could be particularly beneficial for healthcare professionals and patients to identify an optimal and/or personalised course of treatment.
Real-time Guidance
For treatments in which a specific breathing pattern has been identified as optimal for enhancing the delivery and deposition of medication in the respiratory airways, the connectivity function, supported via a mobile app, could provide guiding instructions on when to inhale and exhale using graphics. By relying on pressure change readings transmitted from the AdheResp nebuliser, the data can be combined with the app’s algorithm to understand how best to synchronise breathing with the optimal profile. This would provide feedback to the patient during the treatment for self-adjusting to the expected breathing profile.
Gamification Elements
To support adherence in younger users, the implementation of games in a mobile app could serve as the means to engage with the use of the nebuliser. Pressure changes could be used as a guiding tool in a customised mobile app.
Reminders for Cleaning and Consumable Replacement
Maintenance of the device is an important factor to ensure that patients receive their medication under the optimal delivery conditions. By using the data recorded in the AdheResp nebuliser, it is possible to display reminders in a mobile app for when the medication reservoir should be cleaned, disinfected or when the consumables should be replaced. This patient tool also provides useful information to healthcare professionals and pharmaceutical companies seeking to understand how the patient adheres to the suggested maintenance procedure. Similarly, the effects associated with cases where patients do not adhere to the standard cleaning procedure can also be identified.
Treatment Reminders
Primarily focused on patients, treatment reminders are intended to assist adherence through notifications, which can be configured on the mobile app and linked to the time information transmitted by the AdheResp nebuliser.
Prescriptions and Appointments
While not directly linked to the AdheResp nebuliser, features such as facilitating the issuance of prescriptions and arrangement of appointments for future hospital visits are important tools that benefit patients and healthcare professionals during the course of treatment. These features could indirectly contribute to improved adherence.
“Translating these data into a quantifiable and visual format is necessary for presenting it in a way that patients and healthcare providers can fully benefit from.”
CONNECTIVITY AND COLLABORATION OPPORTUNITIES
As the delivery of connected solutions can extend beyond the medical device features, the collaboration of multiple companies in the development of these products becomes crucial. Smart nebulisers possess integrated Bluetooth modules and other components, enabling the collection and transmission of data. However, translating these data into a quantifiable and visual format is necessary for presenting it in a way that patients and healthcare providers can fully benefit from. To achieve this, mobile app developers who are experienced in creating secure and easy-to-comprehend app platforms have an indispensable role to play.
This type of collaboration requires device manufacturers to share data transmission formats that can be received in mobile apps. Once received, the algorithms created by app developers can process these data to display the desired information. Depending on the function and scope of a mobile app, it may be classified as medical device and require regulatory filing in certain countries and regions.
Similarly, cloud service providers contribute to the exchange of data between all parties that could benefit from it in the process of monitoring adherence. For medical devices, several cloud services providers can ensure the secure transmission and storage of data to protect private information. Although prioritised during development, data privacy remains one of the most controversial issues surrounding the medical IoT, especially in respect to the safety of transmitting data between devices and data ownership once collected. Nonetheless, as new guidelines are being drafted and released more frequently, targeting harmonisation of the overall connectivity environment, the goal is to reduce concerns regarding this aspect as much as is feasibly possible.
Companies such as HCmed Innovations are open to creating collaboration opportunities with app developers and medical cloud service providers to implement the connectivity functions supported by the AdheResp nebuliser. HCmed Innovations’ objective is to establish a network of solutions for future development with pharmaceutical companies, aiming to include connectivity as an effective mechanism to monitor adherence and further improve patients’ treatment outcomes.
OTHER APPLICATIONS AND THE FUTURE OF CONNECTIVITY
Besides the implementation of connectivity in a commercial product, connectivity solutions can also be implemented during clinical trials. Data collection in clinical trials could add value to the outcome of the study, which may not be accurately and effectively recorded without connectivity functions.
Pharmaceutical companies intending to include connectivity in the scope of their trials could benefit from this process, although cybersecurity and the lack of a clearly standardised procedure for the implementation of connectivity in trials remain points to be addressed. Currently, a large number of clinical trials using connectivity have mostly focused on the data aspect or existing therapies, rather the development of a new therapy.
Nevertheless, as technological advancements continue to cement their position in the medical field and new guidelines are released, the future of connectivity is increasingly promising for future treatments, including those in the respiratory field. The AdheResp nebuliser offers an accessible platform for the use of connectivity from early development to commercialisation. Standard and customisable functions comprise options for different indications and target populations, while also incorporating other smart functions, such as breath actuation and device activation, to add value to adherence monitoring.
In the nebuliser space, HCmed Innovations is committed to creating more value by actively collaborating with pharmaceutical companies that aim to incorporate adherence monitoring solutions in new therapies in the push for connectivity implementation, thus assisting patients in their treatment.
REFERENCES
- Makhecha S et al, “Novel electronic adherence monitoring devices in children with asthma: A mixed-methods study”. BMJ Open Respir Res, 2020, Vol 7(1), Article e000589.
- Mosnaim G et al, “Digital Health Technology in Asthma: A Comprehensive Scoping Review”. J Allergy Clin Immunol Pract, 2021, Vol 9(6), pp 2377-2398.
- Zabczyk C, Blakey JD, “The Effect of Connected “Smart” Inhalers on Medication Adherence”. Front Med Technol, 2021, Vol 3, Article 657321.
- Moore A et al, “A randomised controlled trial of the effect of a connected inhaler system on medication adherence in uncontrolled asthmatic patients”. Eur Respir J, 2021, Vol 57(6), Article 2003103.