To Issue 130
Citation: “Interview with Natalia Servol, Head of Unither’s Ophthalmic Business”. ONdrugDelivery, Issue 130 (Mar 2022), pp 17–18.
In this interview, Natalia Servol, Head of Unither Pharmaceuticals’ Ophthalmic Business, speaks about the company’s equipment for compounding and fill-finish of preservative-free products into multidose bottles, part of Unither’s unique and innovative preservative-free multidose offering for partners.
“The key to successfully creating relevant ophthalmological
products lies in sharing ideas with partners.”
Q What equipment do you currently use for ophthalmic products?
A We have a considerable inventory of manufacturing equipment and versatile tank systems allowing us to work on industrial machines, under GMP conditions, with batch sizes from 3 L to 2,000 L. We additionally offer a variety of ophthalmic moulds going from 0.25 mL to 1.0 mL. We can compound and fill various types of formulation, including solutions, emulsions and gels.
Q How do you answer to your customers’ needs such as regulatory and manufacturing requirements?
A I believe that the key to successfully creating relevant ophthalmological products lies in sharing ideas with partners.
Every Unither manufacturing plant has a dedicated R&D team and pilot workshop for the internal development of customer projects. If required, some works can be provided by or executed with the participation of our innovation and development centre in Bordeaux, France, or the back-up manufacturing plant. Our customers benefit from our international R&D and industrialisation footprint end to end, from early-stage work to commercial manufacturing.
Unither has extensive experience of working in a truly international environment, both for project management with partners, and commercial supply. We meet international quality and regulatory standards and are approved by the EU EMA, the US FDA, Brazil’s ANVISA, the Korean MFDS, China’s MoH and many others.
Q What ophthalmic technologies does Unither offer?
A Unither’s industrial engineering offering comprises three main technologies for sterile manufacturing of ophthalmic products: blow-fill-seal (BFS) in single-unit vials, preservative-free multidose (PFMD), and common multidose (MD) for products with preservatives.
We are always innovating solutions to meet our customers’ requirements and we’re always aware that ultimately this equates to meeting patients’ needs. By building the industrial synergy between two main technologies for preservative-free ophthalmic products, BFS and PFMD, we believe we’re truly providing our customers with tailored, sustainable solutions, and patients with products that will enhance their quality of life.
This has been the driving force for our creation and realisation of an innovative PFMD manufacturing line.
Q Can you tell us more about the PFMD manufacturing line?
A Today, we are proud to introduce to you our innovative PFMD line for aseptic manufacturing of ophthalmic products (Figure 1).
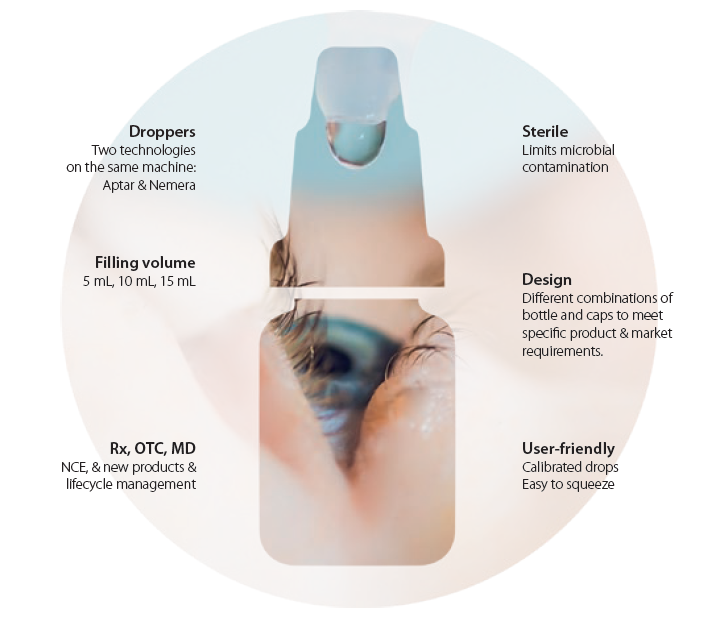
Figure 1: Unither’s innovative PFMD manufacturing line is the embodiment of 25 years of know-how.
“Today, we are proud to introduce to you our innovative PFMD line for aseptic manufacturing of ophthalmic products.”
We are especially proud and delighted with this achievement, a cutting-edge PFMD line that is the embodiment of more than 25 years of sterile know-how. It represents the result of uncountable hours of work by our teams together with external suppliers and customers.
By creating a synergy with unit-dose vials, the PFMD presentation acts as a complement to BFS and gives patients continuous security and product quality throughout their treatment period.
Q How did you come up with this concept?
A The new PFMD offering at Unither came about as a result of internally generated innovation and ideas, from our customers’ feedback, and from our constant drive towards meeting patient needs.
As of today we work with technologies from two major players in the ophthalmic space: Aptar Pharma and Nemera. Looking to the near future, our patient-oriented philosophy means that we foresee novel tailored solutions being added to our offering in the near future.
Q What is coming next for Unither?
A We have plenty of exciting projects underway and on the horizon! We’ve recently taken a strategic decision to transform our Bordeaux R&D site into a Center of Excellence for Ophthalmology R&D, and the entire team is hard at work on this project.
We continue to create innovative solutions together with our partners. Among many novel initiatives we’re supporting, one that I’d like to mention here is CureCall (Paris, France), a start-up specialising in the monitoring of chronic ocular diseases – a user-friendly solution for doctors and patients.
We foresee great challenges in the ophthalmic field and we do believe that vision science can overcome them by collaboration and free sharing of ideas within interdisciplinary teams. Our credo is be open-minded.
Unither will be attending numerous specialised events over the coming months, including the ARVO 2022 Annual Meeting (Denver, CO, US, May 1–4, 2022), and would welcome the opportunity to welcome new contacts to its booth.

