Citation: Mueller-Albers J, “The Secrets of Solubility Enhancement Success with EUDRAGIT® Carriers”. ONdrugDelivery, Online, September 13, 2021.
Jessica Mueller-Albers discusses how Evonik’s EUDRAGIT® functional polymers can enhance drug solubility and permeability, with a focus on carriers, screening tools and manufacturing processes.
“Poly(meth)acrylates have been used for many years in pharmaceutical dosage forms in diverse applications such as for coatings, granulation and tableting. They are synthetic polymers, which ensures low batch-to-batch variability, compared with natural type polymers.”
Find out more from Evonik experts in the webinar Solving solubility challenges with EUDRAGIT® functional polymers on Wednesday, September 22, 2021 at 10:00 a.m. EDT/ 4:00 p.m. CEST.
A successful pharmaceutical product is well-designed and manufactured consistently to ensure it fulfils its intended purpose. In general, both the properties of a drug substance and the choice of polymer affect the formulation and process design of a pharmaceutical, especially its bioavailability, stability, and manufacturability. One very important property of a drug substance is its aqueous solubility.
Almost half of currently marketed drug products, and around 90% of those in clinical development are associated with poor drug solubility or permeability. One way of overcoming poor solubility of drugs is by using functional polymers.
POLY(METH)ACRYLATE POLYMERS
Poly(meth)acrylates have been used for many years in pharmaceutical dosage forms in diverse applications such as for coatings, granulation and tableting. They are synthetic polymers, which ensures low batch-to-batch variability, compared with natural type polymers. Poly(meth)acrylates have a very narrow specification and are monographed in the European Pharmacopoeia, the USP/NF and the Japanese Pharmaceutical Excipients. From Evonik’s offering of EUDRAGIT® functional polymers, 10 EUDRAGIT® E, EUDRAGIT® L 100, EUDRAGIT® L 100-55 and EUDRAGIT® FS have shown the strongest solubility enhancing performance for a wide range of APIs (see Table 1).
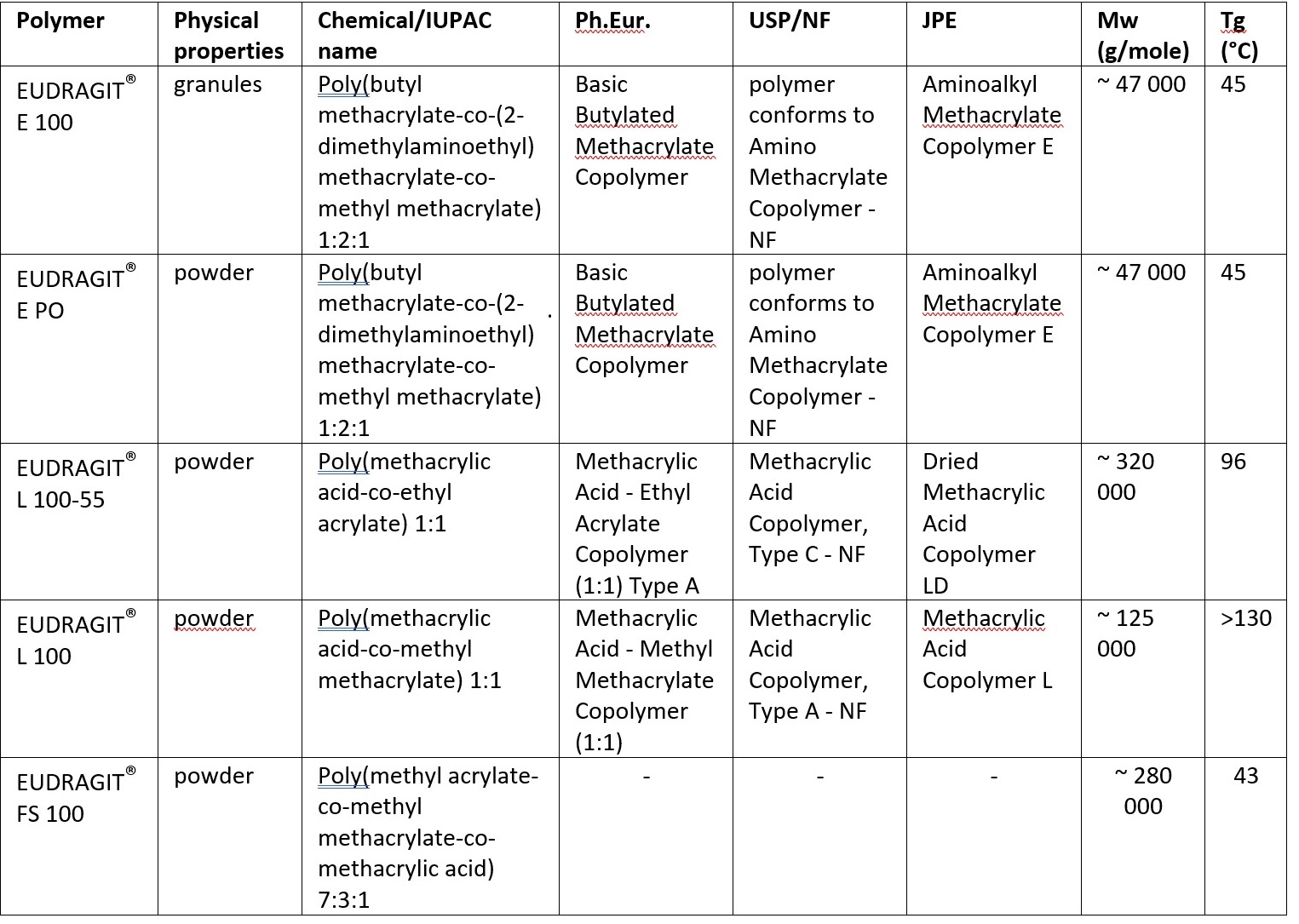
Table 1: Chemical name, compendial compliance and properties of poly(meth)acrylates.
“It is important to consider that the characteristics of a specific excipient – in this case the stabilising polymer – always have to be assessed in relation to the intended functionality, performance and shelf life.”
The most important function of the polymer is to stabilise the amorphous drug in the formulation so that a higher solubility of the amorphous form can be exploited. The molecular weight has an impact because a high molecular weight leads to a higher glass transition temperature (Tg). Furthermore, the molecular weight is directly related to the intrinsic viscosity. A high-molecular-weight polymer forms a high viscosity diffusion boundary layer around solid dispersion molecules. Also, polymers with a high Tg are good stabilising carriers as they reduce molecular mobility. However, it is important to consider that the characteristics of a specific excipient – in this case the stabilising polymer – always have to be assessed in relation to the intended functionality, performance and shelf life.
EUDRAGIT® polymers have been widely used for the formulation of amorphous solid dispersions (ASDs). EUDRAGIT® L 100 and EUDRAGIT® L 100-55 enhance solubility through ASD formation, protect drugs from recrystallisation in gastric fluid and delay supersaturation until the drug reaches the upper small intestine so that absorption is maximised.
Figure 1: Miniaturised screening tool developed by Evonik for the selection of the best carrier for poorly soluble drugs using solvent-based systems such as spray-drying.
CHOICE OF THE RIGHT CARRIER
Selecting the right carrier for solubility enhancement of poorly soluble drugs requires screening of a wide range of polymers or polymer combinations. There is no one-size-fits-all solution because drug ratios and the addition of further excipients must be individually adapted. Screenings require larger quantities of API, as well as time and manual effort for the experiments. Evonik has developed a miniaturised screening tool (Figure 1), which can help identify the best formulation basis while requiring only tiny quantities of API. This is particularly important in the early stages of formulation development when only small amounts of API are available.
MANUFACTURING TECHNOLOGIES
Spray drying and melt extrusion are the technologies that are still the most frequently used and promoted today. Hot melt extrusion (HME) can be used for APIs with a low melting point, and spray drying is suitable for drugs with a good solubility in organic solvents. For all originator products approved by the FDA, spray drying and melt extrusion are the main technologies used. However, other simple methods such as top-spray granulation is being used in the generics markets, where cost pressure and patent complexity is very high. The principle of solubility enhancement via top-spray granulation involves dissolving an API and carrier in an adequate solvent and then displacing the solids on inert starter particles or cores such as sugar spheres or mannitol particles.
CONCLUSION AND OUTLOOK
“EUDRAGIT® can be used in spray-drying and hot melt extrusion but can also increase solubility of poorly soluble drugs when used in simple top-spray granulation processes.”
EUDRAGIT® polymers are excellent carriers for the formation of amorphous solid dispersions due to their superior solubilisation and stabilisation properties. In terms of manufacturing, EUDRAGIT® can be used in spray-drying and hot melt extrusion but can also increase solubility of poorly soluble drugs when used in simple top-spray granulation processes. When examining the whole process, we have seen that early structured excipient and formulation screening shortens time to market and reduces cost. Our research team at Evonik has gathered significant experience in amorphous solid dispersions over the last two decades and uses this to offer our customers well-designed and manufactured oral excipients that demonstrate high performance in a diverse range of processes to formulate ASDs.
Find out more from Evonik experts in the webinar Solving solubility challenges with EUDRAGIT® functional polymers on Wednesday, September 22, 2021 at 10:00 a.m. EDT/ 4:00 p.m. CEST.

