To Issue 147
Citation: Suzuki T, “OXYCAPT: Superior Primary Containers for Biologics and Gene and Cell Therapies” ONdrugDelivery, Issue 147 (May 2023), pp 52–56.
Tomohiro Suzuki reviews the advantages that OXYCAPT™, Mitsubishi Gas Chemical’s multilayer material for vials, offers to biologics and gene and cell therapies, and discusses the results of recent tests into low-temperature storage and dimethyl sulfoxide resistance.

Figure 1: OXYCAPT multilayer plastic vial.
OXYCAPT™ is a multilayer plastic vial developed by Mitsubishi Gas Chemical (MGC) that offers a number of advantageous qualities as a primary drug container (Figure 1). MGC continuously conducts tests to confirm OXYCAPT’s excellent properties, including:
- Excellent oxygen and ultraviolet (UV) light barrier
- Strong water vapour barrier
- Very low extractables
- High pH stability
- Low protein adsorption and aggregation
- High transparency
- High break resistance
- Easy disposability
- Lightweight material.
At present, the company is conducting additional studies into extractables, container closure integrity at cryogenic temperature and other properties, the results of which will be shared at the next opportunity.
In recent news regarding OXYCAPT Syringe, MGC signed a LOI with Becton Dickinson (BD) in May 2022 and have started earnest discussions to apply our multilayer technology to next-generation PFS for biologics. Therefore, we have tentatively stopped introducing the current version of OXYCAPT Syringe to customers. We believe this collaboration will be helpful for the pharmaceutical companies to safely develop novel & sensitive future drugs.

Figure 2: Multilayer structure of OXYCAPT.
“The oxygen barrier of an OXYCAPT-P vial is about 20 times better than that of a COP monolayer vial.”
OXYCAPT OVERVIEW
OXYCAPT consists of three layers – the drug contact layer and the outer layer are made of cyclic-olefin polymer (COP), and the oxygen barrier layer is made of MGC’s novel polyester (Figure 2). One variety of OXYCAPT, OXYCAPT-P, provides an excellent oxygen barrier. For example, the oxygen barrier of an OXYCAPT-P vial is about 20 times better than that of a COP monolayer vial (Figure 3).
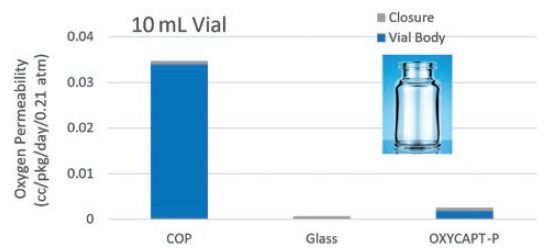
Figure 3: Oxygen permeability comparison of a typical COP, glass and OXYCAPT-P.
MGC recently obtained a report on the environmental impact of glass and plastic containers for medical use from a Japanese research company. The report shows that plastic containers for medical use are much more environmentally friendly compared with glass containers. For example, the carbon footprint, nitrogen oxides (NOx) emissions, sulfur oxides (SOx) emissions and water consumption associated with plastic containers for medical use are several times smaller than those of their glass equivalents.
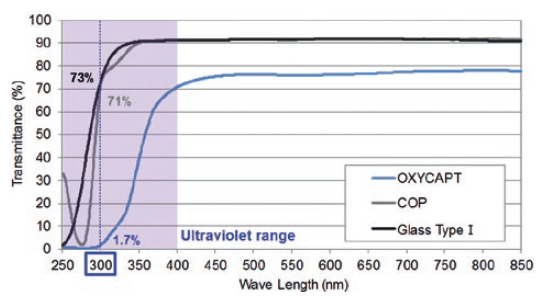
Figure 4: UV light transmittance comparison of a typical COP, Type 1 glass and OXYCAPT.
Furthermore, OXYCAPT provides an excellent UV barrier. While about 70% of 300 nm UV light transmits through glass and COP, only 1.7% transmits through OXYCAPT (Figure 4). MGC has confirmed that this feature contributes to the stability of biologics.
While OXYCAPT cannot reach the performance of glass with respect to acting as a water vapour barrier, its properties are similar to those of COP, which has been used for injectable drugs for a long time. This means that OXYCAPT easily meets the requirements of a water vapour barrier set out by the International Council for Harmonisation (ICH) guidelines.
Studies have shown an extremely low level of extractables from OXYCAPT. One study was conducted to confirm the levels of volatile, semi-volatile and non-volatile impurities from OXYCAPT. Water and four solutions (50% ethanol, sodium chloride, sodium hydroxide and phosphoric acid) were selected and impurities were measured by gas chromatography mass spectrometry (GC-MS) and liquid chromatography-UV spectroscopy-mass spectrometry (LC-UV-MS) after 70 days at 40°C. Compared with the control, impurities were not detected in the OXYCAPT containers. A second study confirmed that inorganic extractables levels from OXYCAPT were similar to those from COP, which is well known for being an extremely pure polymer with a better extractables profile than Type 1 glass (Figure 5). Lower levels of inorganic extractables are known to contribute to better pH stability in drug products.
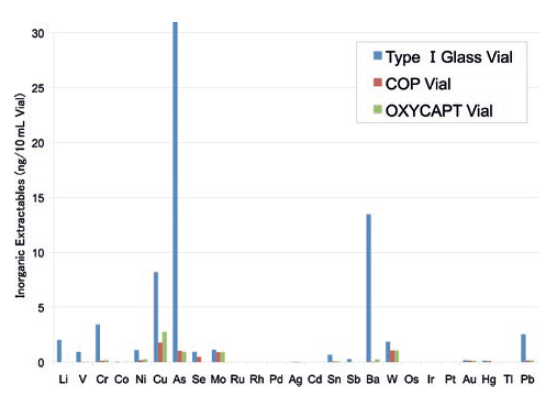
Figure 5: Comparison of inorganic extractables found in COP, Type 1 glass and OXYCAPT.
The OXYCAPT vial is produced by co-injection moulding technology. MGC has also developed inspection methods for testing the oxygen barrier layer. All the containers are fully inspected by state-of the- art inspection machinery.
MGC can offer bulk vials and ready to- use (RTU) vials, with its RTU products provided in standard nest and tub formats. The nest and tub are mainly sterilised using gamma rays. There are 2, 6, 10 and 20 mL variants for vials (Table 1). MGC is willing to provide samples for initial testing free of charge.
| ISO Vial | Height (mm) |
Outer Diameter of Body (mm) |
Outer Diameter of Crown (mm) |
Inner Diameter of Crown (mm) |
Option |
| 2R (2 mL) | 35 | 16 | 13 | 7 | Bulk or RTU |
| 6R (6 mL) | 40 | 22 | 20 | 12.6 | Bulk or RTU |
| 10R (10 mL) | 45 | 24 | 20 | 12.6 | Bulk or RTU |
| 20R (20 mL) | 55 | 30 | 20 | 12.6 | Bulk or RTU |
Table 1: MGC’s OXYCAPT product portfolio.
Each polymer meets the requirements of US Pharmacopeia (USP) regulations USP<661>, USP<87> and USP<88>, as well as those of the European Pharmacopoeia, and has been filed in the US FDA’s drug master file (DMF). The vials are also compliant with each pharmacopoeia and have been filed in the DMF.
“Some drug developers have recently started evaluating the OXYCAPT vials for their gene and cell therapies; the RTU vial is sterilised by gamma radiation, making it ideal for protein-based drugs.”
The primary target market for OXCAPT is the therapeutic application of biologics. As mentioned in ICH Q5C (Stability of Biotechnological/Biological Products), oxidation is one of the causes of protein instability. As such, the oxygen and UV barrier properties of OXYCAPT will definitely contribute to the stability of biologics stored within. Furthermore, some drug developers have recently started evaluating the OXYCAPT vials for their gene and cell therapies; the RTU vial is sterilised by gamma radiation, making it ideal for protein-based drugs.
OXYCAPT AT VERY LOW TEMPERATURES
To verify the suitability of OXYCAPT for drugs stored at very low temperatures, MGC carried out studies using OXYCAPT vials. As customers often ask about the durability of OXYCAPT at low temperatures, MGC conducted quick defrosting and dropping tests. Firstly, the vials were stored in a freezer at -80°C for one day. After being removed from the freezer, the frozen vials were immediately dipped into hot water (40°C) for 15 minutes. No breakage, leakage or layer separation was detected in any of the vials (Figure 6).
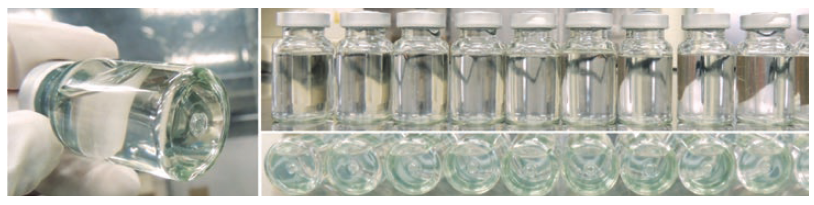
Figure 6: No breakage, leakage or layer separation was found after the quick defrosting test (-80°C to 40°C).
A further test was conducted where the vials were stored in a freezer at -80°C for one-week, six-month and 24-month periods. After being removed from the freezer, the vials were immediately dropped onto a steel plate from a height of 1.5 m. No breakage or leakage was detected in any of the vials for any length of time in cold storage.

Figure 7: Broken COP vial after drop test following storage at -180°C.
The same dropping test was conducted using OXYCAPT and COP monolayer vials that had been stored at approximately -180°C, as regenerative medicines such as gene and cell therapies are often preserved in liquid nitrogen gas-phase freezers. After being removed from the liquid nitrogen gas-phase freezer, the vials were immediately dropped to a steel plate from a height of 1.5 m. Although eight of the COP-monolayer vials were broken (Figure 7), no breakage or leakage was detected in any of the OXYCAPT vials (Table 2). For clarification, as it was considered obvious that glass vials would shatter as a result of these tests and present a safety risk to the experimenters, glass was excluded from the test.
| OXYCAPT Vial | COP Monolayer Vial | |
| Breakage | 0/20 | 8/20 |
| Leakage* | 0/20 | 8/20 |
* Dropped vials were stored at room temperature until the frozen water was defrosted and then leakage was observed.
Table 2: Breakage and leakage from COP and OXYCAPT vials stored at -180°C.
DMSO RESISTANCE
Dimethyl sulfoxide (DMSO) is often used as a cryoprotectant for gene and call therapies because it has been demonstrated that it can prevent the intracellular freezing that causes cell death. As MGC is often asked by customers about OXYCAPT’s DMSO resistance, the company conducted some related studies. OXYCAPT 10R vials with polytetrafluoroethylene (PTFE) stoppers were filled with 10 mL of 10% or 20% by weight DMSO solutions and stored at 40°C for 70 days. Then, volatile impurities were measured by headspace gas chromatography/mass spectrometry (HS-GC/MS) and gas chromatography/mass spectrometry (GC/MS) while non-volatile impurities were measured by ultra-high-performance liquid chromatography/ion trap Fourier transform mass spectrometry (UHPLC/IT-FT-MS). No impurities derived from OXYCAPT vials were detected in either the 10% or the 20% by weight DMSO solutions (Table 3).
| Analytical method | Target materials |
OXYCAPT-P Vial | |||
| 10wt% DMSO (aq) | 20wt% DMSO (aq) | 20wt% DMSO (aq) | |||
| HS-GC/MS | Volatile impurities |
ND from vial* | ND from vial* | ND from vial* | |
| GC/MS | ND | ND | ND | ||
| UHPLC/ IT-FT-MS |
Positive mode | Non-volatile impurities | ND from vial* | ND from vial* | ND from vial* |
| Negative mode | ND | ND | ND | ||
* Two kinds of impurities were detected from both OXYCAPT and Type 1 glass. As the impurities were of the same kind, it is believed that these were derived from the closures.
Table 3: DMSO resistance of OXYCAPT.
CONCLUSION
These latest results have contributed to MGC’s ongoing studies verifying OXYCAPT’s superior properties for biologics and gene and cell therapies. In addition to the advantages of COP, such as a strong water vapour barrier, high break resistance, very low extractables and low protein adsorption, OXYCAPT also provides a strong oxygen and UV barrier. MGC believes that OXYCAPT offers a multitude of benefits to the rapidly growing field of biologics and gene and cell therapies.


