To Issue 133
Citation: Cordier S, Holterboch V, Verger E, “Premiumcoat® Solutions: Helping Control the Level of Silicone to Protect Sensitive Therapeutics”. ONdrugDelivery, Issue 133 (May 2022), pp 48–52.
Sébastien Cordier, Vincent Holterboch and Estelle Verger discuss how coated elastomer components, such as Aptar Pharma’s PremiumCoat®, can help drug developers optimise the balance between component processability and minimising silicone migration into a drug product.
“Whether a vial or PFS, primary packaging must be precisely engineered to protect the drug formulation from outside contamination and degradation throughout its shelf life.”
PROTECTING DRUG FORMULATIONS
From oncolytic drugs to biologics and covid-19 vaccines, injectable drug formulations undergo a long journey before their therapeutic benefits can be brought to patients around the world. A crucial aspect of success on this journey is finding the optimal primary packaging, which plays a critical role in maintaining the stability and efficiency of the drug formulation.
Whether a vial or prefilled syringe (PFS), primary packaging must be precisely engineered to protect the drug formulation from outside contamination and degradation throughout its shelf life. Particulate contamination, faulty container closure integrity, extractables and leachables or other chemical contaminations are some of the many factors that may compromise the API and present a threat to patient safety. Increasing focus is therefore being placed on the importance of minimising the risks related to interaction between a drug formulation and its primary packaging. These considerations become even more acute for sensitive formulations, such as protein-based biologics, which make up a growing proportion of the injectable medicine landscape, and covid-19 vaccines, which leverage advanced formulations of biomolecules.
Aside from glass containers, the only primary packaging components in direct contact with the drug formulation are vial stoppers, PFS plungers and tip closures. In addition to protecting the drug and allowing its easy delivery, these elements must also be easily processable on filling lines, either by pharma companies or contracted manufacturers. Siliconisation, which involves lubricating rubber components with silicone oil, is a necessary process to secure the machinability of stoppers and plungers. It is required to prevent components from sticking together and to reduce friction with filling equipment and other components, thereby limiting the potential for elastomeric particles to be generated during processing. Silicone oil also helps ease the stopper’s application onto the vial and, in the case of syringe plungers, helps reduce the friction with the glass barrel. As such, siliconisation minimises break-loose and glide forces, leading to improved injection system performance when PFSs are activated by healthcare professionals, patients or within an autoinjector.
“PremiumCoat® high-quality components have been proven to reduce extractables and leachables.”
Over the past few decades, the pharmaceutical packaging industry has developed advanced rubber formulations to reduce the quantity of extractables and leachables that may be transferred into the drug formulation. Furthermore, by coating the rubber with fluorinated films, such as ethylene tetrafluoroethylene (ETFE), the amount of extractables and leachables can be further reduced. Studies have reported that the silicone oil applied to the surface of glass syringes and elastomers could migrate into the injectable solution and risk being injected into patients.1 It is therefore important for packaging manufacturers to ensure that silicone levels are kept under control to mitigate the associated risks and help prevent the migration of silicone into the injectable solution.
The result of such advances can be seen in PremiumCoat®, a platform of ETFE film-coated solutions for vials and PFSs developed by Aptar Pharma. These high-quality components have been proven to reduce extractables and leachables, are compatible with standard vial-neck designs, are appropriate for multipiercing applications and demonstrate excellent functional performance on filling lines and during the injection procedure.
Preliminary research conducted by Aptar Pharma, in collaboration with the Institute of Materials Science (Mulhouse, France), used X-ray photoelectron spectroscopy, a technique for analysing the atomic composition of a surface, showed that less silicon may be detected on film-coated surfaces (in the form of silicone oil) than uncoated surfaces.2 Furthermore, Fourier transform infrared spectroscopy (FTIR), a technique for analysing the structure of proteins, demonstrated that model proteins adsorbed on the surface of coated elastomers formed fewer secondary structures (ß-sheets), that are known to be associated with higher chances of aggregation.3,4 Activity assays conjointly demonstrated that proteins adsorbed on coated components were more likely to keep their activity than proteins exposed to naked elastomers.2 Interestingly, further FTIR assays demonstrated that higher levels of silicone were associated with increased formation of ß-sheets.2 Taken together, these results, based on model proteins, suggest that an interplay between PremiumCoat’s coating and reduced amounts of silicone may promote protein stability during storage, further emphasising the need to carefully control the level of silicone oil on closure components.
Aptar Pharma uses a proprietary siliconisation process for its products, including PremiumCoat®. Instead of silicone oil being applied as an emulsion during the washing process, Aptar Pharma’s siliconisation process is carried out during the drying phase of manufacture. The high temperatures involved help make the silicone oil more fluid, easing its transfer onto elastomer components via contact with the wall of the dryer and contact between the products. Because the inside of the stopper cavity does not offer a point of contact, the chance for excess silicone oil to accumulate there is limited. To explore the silicone oil levels associated with PremiumCoat® further, Aptar Pharma conducted a series of detailed tests evaluating the silicone distribution on its products, and comparing them with other ETFE-coated components found on the market.
“The presence of silicone oil is essential for closure components to fulfil their critical function as part of the primary packaging; however, ensuring that it is applied in the smallest possible quantity reduces the risk of silicone contamination, denaturation and aggregation for sensitive biomolecules.”
ACCURATELY MEASURING THE PRESENCE OF SILICONE
Because of the small quantities involved, specialist techniques are required to measure silicone levels accurately. In the tests conducted by Aptar Pharma, the analytical method used was inductively coupled plasma optical emission spectrometry (ICP-OES). In principle, the atoms within a sample are excited by exposure to plasma and the light-emission spectrum produced as the atom recovers its unexcited state is analysed with a spectrometer. Light-emission signatures are directly related to a sample’s atomic composition, thereby enabling the accurate quantification of specific elements, including the silicon atoms that form an integral part of silicone oil. The corresponding silicone quantity was calculated from the atomic mass of silicon and the molecular weight and density of silicone.
PREMIUMCOAT® STOPPERS DISPLAY MINIMAL SILICONE QUANTITIES ON DRUG CONTACT SURFACE
The PremiumCoat® manufacturing process has been optimised across all product formats so that the minimum acceptable silicone quantity is added during the drying step. To measure the amount of silicone across the entire surface of the components, the stoppers were totally immersed in Premisolv, a solvent that maximises the extraction of silicone from the component’s surface as described in figure 1A. The extract was then analysed by ICP-OES and the distribution of silicone expressed as a ratio of the total quantity of silicone on the stopper with regard to the stopper surface. The quantity of silicone that may come into contact with the drug formulation is evaluated following the protocol featured in Figure 1B, and this quantity is expressed as a ratio with regard to the film-coated surface, which is by design the surface that may come in contact with the drug.
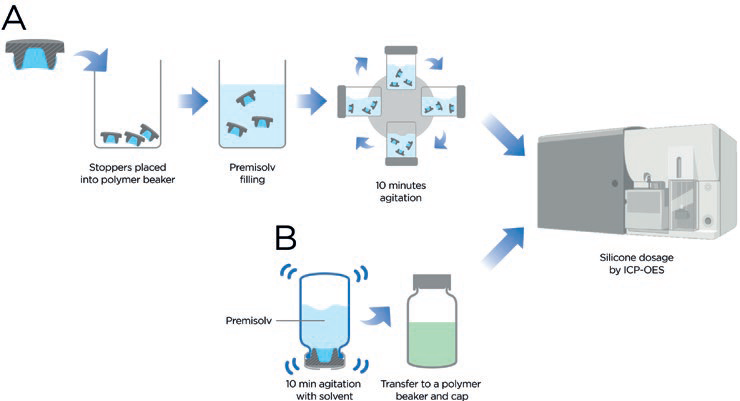
Figure 1: Protocol for measuring the quantity of silicone using ICP-OES. A) The total quantity of silicone is measured by fully immersing stoppers in PremiSolv to extract the silicone oil from the component. The solution is then submitted to ICP-OES for quantifying silicone. B) The quantity of silicone that may be in contact with the drug product is evaluated by inverting a glass vial filled with PremiSolv and closed with the stopper of interest. The solution is then transferred to a polymer vial and analysed by ICP-OES.
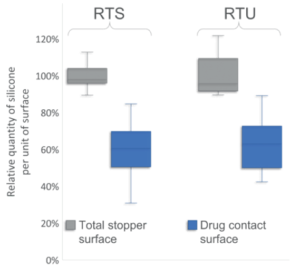
Figure 2: Comparing the distribution of silicone per unit of surface on the total stopper surface and the drug contact surface. For the total stopper surface, three batches were tested with three repetitions per batch and three stoppers per repetition (n=9, total of 27 20 mm stoppers). For the drug contact surface, three batches of 30 stoppers were tested (n=90 20 mm stoppers). Relative quantity of silicone expressed as percentage of average quantity of silicone measured on the total stopper surface. Ready-to-Sterilise (RTS) stoppers were not sterile and Ready-to-Use (RTU) stoppers were gamma-sterilised.
Figure 2 shows that, for both conditions analysed, PremiumCoat® stoppers displayed relatively less silicone per unit of surface at the drug contact surface than on the total surface of the stoppers. For both non-sterilised RTS and gamma-sterilised RTU stoppers, the drug contact surface displays on average 40% less silicone than the rest of the stopper.
This observation is a clear indication that Aptar Pharma manufacturing process optismises the distribution of silicone on the stopper’s surface. The significantly lower quantity of silicone that is exposed to the drug can help preserve its integrity, while the higher quantity of silicone on the rest of the stoppers may promote optimal machinability on customers filling lines.
PREMIUMCOAT® AND OTHER COATED COMPONENTS PERFORM SIMILARLY WITH REGARD TO SILICONE
As biologics and sensitive drugs become more common on the market, other manufacturers have begun supplying ETFE-coated components. The potential issues caused by silicone oil have been raised by the market, and additional surface treatments have been developed to help mitigate the risks linked to the silicone that may be exposed to the drug product. To compare the performance of other ETFE-coated components with that of PremiumCoat®, Aptar Pharma performed additional tests to evaluate the amount of silicone that may migrate into the drug solution for an alternative ETFE-coated stopper, applying the same protocol described in Figure 1B.
In Figure 3, we compare the quantity of silicone extracted from the drug contact surface of PremiumCoat® with the quantity of silicone extracted from the drug contact surface of another type of ETFE-coated stoppers for which additional surface treatments are performed. In the conditions of this test, as performed by Aptar Pharma, PremiumCoat® and the other ETFE-coated stopper displayed similar performances, with all stoppers showing comparable quantities of silicone extracted. Our results indicate that for both Aptar Pharma PremiumCoat® and the other ETFE-coated stoppers, a similar fraction of silicone may come into contact with the drug product.
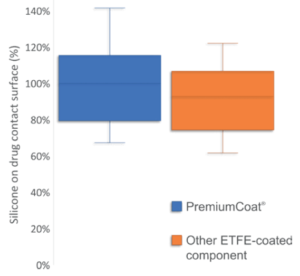
Figure 3: Comparison of the quantity of silicone that may be extracted from the stopper’s drug contact surface of 20 mm, RTU gamma-sterilized PremiumCoat® (n=90) or another 20 mm RTU steam-sterilised ETFE-coated stopper (n=60) . Quantity of silicone expressed as percentage of average value for PremiumCoat® components.
In conclusion, these results demonstrate that both stoppers are susceptible to releasing very low amounts of silicone into the drug solution. It is important to note that PremiSolv is an aggressive solvent and extracts the totality of the silicone from the surfaces it touches. In practical situations, most therapeutic solutions are solubilised in non-aggressive aqueous solutions, minimising the risk of silicone migration into the drug product.
PREMIUMCOAT® HELPS MITIGATE THE RISKS LINKED TO SILICONE OIL
The findings of these tests are important to help inform decisions regarding the choice of optimal primary packaging for sensitive drug developments. Elastomeric closure components introduce several materials, including silicone oil, into the vicinity of the active drug. This opens the door to a range of possible unwanted interactions that have the potential to compromise the potency of a drug and patient safety for injections.
The presence of silicone oil is essential for closure components to fulfil their critical function as part of the primary packaging; however, ensuring that it is applied in the smallest possible quantity reduces the risk of silicone contamination, denaturation and aggregation for sensitive biomolecules. Aptar Pharma has successfully managed to fulfil this objective with its PremiumCoat solutions to allow the early de-risking of packaging concepts for drugs designers.
For manufacturers of biomolecule-based therapeutics, the complex and dynamic interplay between all the variables within the primary container highlights the need for a deeper understanding of the materials and processes involved. Being equipped with this knowledge greatly increases the chances of success when developing a drug, ensuring its long-term integrity and guaranteeing that every patient receives a safe dose.
All presented data are extracted from routine in-process quality checks and MDNFET001.
For more information about PremiumCoat®, visit: www.aptar.com/products/pharmaceutical/premiumcoat-coated-vial-stoppers-and-syringe-plungers.
REFERENCES
- Yu JH et al, “Silicone Oil Droplets Following Intravitreal Bevacizumab Injections”. Am J Ophthalmol Case Rep, 2018, Vol 10, pp 142–144.
- Huang T, “Protein Adsorption and Denaturation In Injectable Devices for Pharmaceutical Applications”. PhD thesis, 2016, Institut de Science des Matériaux de Mulhouse & Aptar Pharma.
- Dong A et al, “Infrared Spectroscopic Studies of Lyophilization- and Temperature-Induced Protein Aggregation”. J Pharm Sci, 1995, Vol 84(4), pp 415–424.
- Fink AL, “Protein Aggregation: Folding Aggregates, Inclusion Bodies and Amyloid”. Fold Des, Vol 3(1), pp R9–R23.

