To Issue 183
Citation: Barron L, “The Challenges in Developing and Manufacturing Custom Ophthalmic Injector Devices”, ONdrugDelivery, Issue 183 (Mar 2026), pp 38–44.
Dr Lorna Barron sets out the standards that must be met for effective drug delivery to the eye, characterising both old and new delivery routes that may be used to address the unique challenges of ophthalmic disease.
The global demand for ophthalmic drugs continues to rise, driven by aging populations,1 increasing prevalence of chronic eye diseases and improved access to healthcare worldwide. Conditions such as age-related macular degeneration (AMD), diabetic retinopathy and glaucoma are being diagnosed earlier and treated more aggressively than ever before. As a result, pharmaceutical companies are investing heavily in new formulations and delivery methods to meet both clinical needs and patient expectations.
“DIFFERENCES IN VISCOSITIES, TARGET INJECTION VOLUME, FORMULATION PROPERTIES AND CLINICAL WORKFLOW REQUIRE TAILORED SOLUTIONS.”
With the rapidly diversifying pipeline of ophthalmic treatments, including biologics, high-viscosity formulations, sustained-release systems and gene therapies, significant delivery challenges have arisen that legacy injector platforms are not designed to accommodate. Differences in viscosities, target injection volume, formulation properties and clinical workflow require tailored solutions. This need is reflected in market forecasts, with the global ophthalmic drug delivery device market projected to grow from approximately US$16.5 billion (£12.2 billion) in 2024 to $37 billion by 2033.2
There are several targets for ophthalmic drug administration, including topical, intravitreal and subretinal. Each have their own challenges that require unique solutions (Table 1).
| Route | Advantages | Disadvantages |
| Topical (eye drops) | Non-invasive, easy. | Low bioavailability due to corneal barrier and tear drainage. |
| Intravitreal injection | Direct delivery to back section of the eye (posterior segment). |
Invasive. Risks such as infection. Clinician must administer. Frequent repeats mean high treatment burden. |
| Subretinal injection | Direct and localised target for retinal therapies. Appropriate for cell and gene therapies. |
Invasive, requires complex procedure by highly skilled surgeon, risk of severe complications: retinal detachment, haemorrhage, etc. |
| Suprachoroidal injection | Targets posterior segment while limiting systemic exposure. Does not require full operating theatre. | Targeting of retina is less precise. Not as well established. |
Table 1: Several different delivery routes for ophthalmic drugs, each with advantages and disadvantages.
TOPICAL DRUG DELIVERY
Topical drug delivery, most commonly via eye drops, remains the most widely used non-invasive approach for treating ocular conditions, yet its effectiveness is fundamentally constrained by ocular anatomy and physiology. Only a small fraction of an administered drop is retained on the ocular surface, as rapid tear turnover, reflexive blinking and limited tear volume lead to swift drug loss within minutes. In addition, drugs must traverse multiple corneal barriers with differing hydrophobicity and permeability characteristics, significantly limiting penetration and overall bioavailability. These factors often require frequent and higher dosing, increasing the risk of local and systemic side effects and contributing to poor patient adherence – particularly among elderly or physically limited populations.3
Researchers are actively developing new approaches to overcome these limitations, with key aims including increased residence time on the ocular surface, enhancement of tissue penetration and sustained drug release. Formulation innovations such as mucoadhesive polymers and in situ gelling systems can prolong contact with the precorneal environment and reduce rapid tear clearance. Additionally, advanced carrier systems including nanoparticles, liposomes and micelles can be used to improve corneal permeation and protect drugs from degradation. Finally, drug-eluting contact lenses and ocular inserts may provide sustained-release solutions without frequent dosing.
INTRAVITREAL INJECTIONS
Intravitreal injections are one of the most commonly performed ophthalmic procedures worldwide. These injections are typically administered in an outpatient clinical setting and involve the insertion of a needle through the sclera to inject directly into the vitreous humour, the gel-like substance that fills the central cavity of the eye. This delivery route allows drugs to bypass the barriers experienced by topical delivery and reach the posterior segment of the eye. Today, intravitreal injections of anti-vascular endothelial growth factor agents are an essential treatment for millions of patients with retinal vascular diseases, such as wet AMD and diabetic retinopathy.
Intravitreal injections, while clinically effective for delivering drugs to the posterior segment of the eye, also present several limitations and risks. They are inherently invasive, risking serious complications such as sight-threatening retinal detachment or endophthalmitis. Some procedures, such as intravitreal corticosteroid injections, can lead to increased intraocular pressure (IOP) due to increased outflow resistance of aqueous humour in the trabecular meshwork. Often, this acute effect may subside after injection; however, chronic IOP increases may occur with multiple injections, potentially leading to glaucoma over time. Many intravitreal drugs have relatively short half-lives, requiring repeat monthly or bimonthly injections. This high treatment burden places strain on patients, caregivers and healthcare systems and is associated with reduced adherence over time.4
Conventional syringe-based injections offer limited control over injection force, flow rate and drug distribution, which can be problematic regarding dosing variability, reflux at the injection site and cumulative ocular trauma. Sustained-release intravitreal implants and reservoir systems (such as port delivery systems) are being developed to reduce injection frequency and treatment burden, offering controlled long-term release while potentially lowering cumulative procedural risk and improving adherence.5–7
One such example is Contivue® a port delivery platform containing Susvimo®, a ranibizumab injection by Roche (Basel, Switzerland). This refillable intravitreal implant continuously delivers a customised formulation of ranivizumab over extended periods, significantly reducing the frequency of intravitreal injections required for chronic retinal diseases including neovascular (wet) AMD and diabetic retinopathy. This offers continuous delivery with just one refill every nine months for appropriate patients. The device is surgically implanted into the eye during a one-time outpatient procedure, with ancilliary devices used to fill, insert, refill and remove the implant.8,9
“THIS DIRECT DELIVERY FACILITATES HIGH LOCAL DRUG CONCENTRATIONS, RAPID UPTAKE BY TARGET CELLS AND MINIMAL SYSTEMIC EXPOSURE, MAKING IT ESPECIALLY VALUABLE FOR GENE AND CELL THERAPIES.”
SUBRETINAL INJECTIONS
Subretinal injection delivers therapeutic agents directly into the space between the photoreceptors and retinal pigment epithelium, providing high-precision targeting of cells central to many retinal degenerative diseases, such as retinitis pigmentosa, AMD and inherited retinal disorders. This direct delivery facilitates high local drug concentrations, rapid uptake by target cells and minimal systemic exposure, making it especially valuable for gene and cell therapies where precise placement determines treatment efficacy. Moreover, the immune-privileged nature of the subretinal space can reduce immune reactions against viral vectors or transplanted cells compared with other routes.10,11
However, subretinal injections come with significant challenges. The procedure is highly technical and invasive, typically requiring a pars plana vitrectomy and creation of a subretinal bleb, which can itself pose risks for retinal tears, detachment, haemorrhage and cataract formation. Needle placement is difficult due to the tiny target space, and even small lateral movements can widen the retinotomy and promote reflux of the therapeutic agent into the vitreous, reducing delivery efficiency. Variability in injection speed and plunger movement can significantly influence the delivered volume and therapeutic effect, leading to dependence on the skill of the practitioner for manual devices such as standard intravitreal injection syringes. Additionally, the retinal cells may be subjected to high stress from a subretinal bleb that is created too quickly. Therefore, specialist surgical training and intraoperative imaging, such as optical coherence tomography (OCT), are required and robotic assistance is increasingly needed to improve precision and reduce variability.12,13
To address these challenges, researchers are exploring advanced techniques and devices, including ultra-fine cannulas, intraoperative OCT guidance and robot-assisted platforms, with the aim to standardise delivery, control injection depth and pressure, suppress tremors and reduce complications (Figure 1). These advances could make subretinal delivery both safer and more reproducible for gene, cell and other localised therapies.11,14

Figure 1: A generic device used to inject drug in the suprachoroidal space.
DEVICE DEVELOPMENT AND MANUFACTURING CHALLENGES
There is a clear need for delivery devices to go beyond conventional eye drop bottles and syringes. However, to translate emerging methods into the clinic, several important challenges must be overcome, including complex engineering needs, human factors and regulatory considerations.
Ophthalmic devices may require complex engineering to deliver new therapies. Formulations including carrier systems, such as nanoparticles or liposomes, may be sensitive to shear stress, requiring careful flow analysis of systems. Dosing accuracy on the microlitre scale is also essential, requiring control of mechanical tolerances, force feedback and fluid dynamics.
As the trend grows for high-viscosity biologics, there is a need for greater injection forces, which must be carefully controlled to ensure safe intraocular delivery. Additionally, sustained-release solutions, such as drug-eluting contact lenses or ocular inserts, must ensure predictable release kinetics.
Incorporating human factors into device development is also essential to reduce the risk of user error and to increase patient adherence. For topical applications, in situ gels, viscous formulations and mucoadhesives must be precisely dispensed in small, repeatable volumes without relying on patient technique. Devices must be intuitive, comfortable and suitable for elderly or physically limited patients, minimising the need for precise hand-eye co-ordination or frequent dosing.
Sanner conducts comprehensive human factors and usability studies to ensure that devices are designed around real-world users, environments and workflows. The company’s studies evaluate critical user interactions, identify potential use-related risks early in development and inform design refinements that improve safety, usability and reliability. For more complex intraocular delivery, clinicians must be able to operate devices safely and consistently, and variation in clinicians’ skill levels should not adversely affect the operation of the device. Through formative and summative human factors testing, Sanner helps to ensure that devices support consistent clinical performance, reduce training burden and meet regulatory expectations for usability and risk mitigation.
Finally, regulatory requirements are a necessary part of device development. The eye is a vulnerable organ of the body, with injuries causing life-changing consequences. As these devices come into direct contact with ocular tissues, any contamination can cause adverse reactions ranging from irritation, infection or even vision impairment. Ophthalmic products and devices must therefore follow strict regulatory requirements. Different countries have various regulatory frameworks that account for factors such as packaging, sterilisation, quality control and adverse-event reporting. Additionally new ophthalmic devices often combine drug, device or biologic components, which must be designed with regulatory compliance in mind from the start. Robust safety and efficacy data are required before market approval can be obtained, with extensive documentation and validation. These hurdles can extend development timelines and increase R&D costs, especially for novel devices.
“SANNER HAS EXTENSIVE EXPERIENCE IN USING MULTIPHYSICS MODELS TO INFORM DEVICE DESIGN AND FACILITATE OPTIMISED PROTOTYPES AT AN EARLY STAGE OF DEVELOPMENT.”
COMPUTATIONAL MODELLING AS A DEVICE DEVELOPMENT TOOL
Computational modelling is one tool that can be used to facilitate the design process. Computational fluid dynamics (CFD) modelling of both flow within an injector system and flow within the eye can provide insights such as shear stress applied to carrier systems, increases in intraocular pressure for different flow conditions or the injection forces required for different formulation viscosities. Release kinetics for different positioning or composition of ocular inserts can also be calculated using mass transport or multiphysics models to inform designs at an early stage.
The benefit of modelling techniques such as finite element analysis (FEA) and CFD is that they allow for efficient predictive testing of device performance under different designs and conditions. The impact of anatomical variation between patients on the biomechanics of the system can be examined by simple parameter changes in a model. This reduces the need for physical prototyping which, in turn, can shorten development timelines, lower costs and identify potential failure modes prior to clinical evaluation. Sanner has extensive experience in using multiphysics models to inform device design and facilitate optimised prototypes at an early stage of development.
MANUFACTURING
Once a device design is finalised, manufacturing custom ophthalmic devices at scale introduces additional challenges. One challenge of manufacture is that ophthalmic devices target micro-scale anatomical features. Therefore, they often involve small components with tight tolerances and pristine surface finishes, which must maintain consistency across production lots. Sanner has deep expertise in precision injection moulding for pharmaceutical devices. Additionally, its new site in Germany offers cleanroom manufacturing, which is key to producing ophthalmic device parts.
Materials selection is also a major consideration, as the materials used for the device must be compatible with the drug formulation to ensure that aggregation, adsorption or degradation do not occur. Leaching of chemicals or particle generation from silicone oil coatings must also be avoided, and the device materials and drug formulation must also be able to withstand sterilisation. Wall thicknesses and aspect ratios may approach the limits of what can reasonably be manufactured for materials that meet all other requirements for a specific formulation.15
In addition, to be used for clinical drug use, sterile manufacturing must be carried out to meet regulations, pass quality control tests and maintain consistency throughout the entire manufacturing process. Depending on the device and formulation, the need for aseptic processing may arise. Terminal sterilisation of filled drug products is often the preferred approach of regulatory bodies; however, for many ophthalmic therapies, including cell and gene therapies, these sterilisation processes can cause degradation of the product. Sanner is a strong choice to help clients develop sterilisation concepts, and the company works closely with vetted external partners to achieve this.
“SUCCESSFULLY MOVING CUSTOM OPHTHALMIC INJECTORS FROM CONCEPT TO MARKET REQUIRES A PARTNER WITH DEEP EXPERTISE ACROSS DESIGN, DEVELOPMENT, REGULATORY PATHWAYS AND MANUFACTURING EXECUTION.”
Finally, scale-up of these processes can also be a challenge. Novel injection devices may rely on bespoke parts and materials that may have limited sources or long lead times. Careful planning is needed to reduce strain on the supply chain, as any delay in production may inflate costs.
CHOOSING THE RIGHT PARTNER
Due to the unique hurdles in development and manufacture of new ophthalmic injection devices, it is essential to choose a partner with the right experience. Successfully moving custom ophthalmic injectors from concept to market requires a partner with deep expertise across design, development, regulatory pathways and manufacturing execution (Table 2).
| Challenge | Capabilities | Example Application |
| Complex engineering tailored to new therapies | Computational modelling (e.g. CFD, FEA, multiphysics modelling) to optimise flow, injection forces, shear stress and release kinetics prior to prototyping. |
Device design for delivery of a shear-sensitive high-viscosity nanoparticle suspension. |
| Microlitre-scale dosing accuracy | Precision engineering with tight mechanical tolerances, practical testing of forces and fluid dynamics analysis. | A subretinal injector that requires high dose accuracy. |
| Sustained-release systems | Computational modelling to predict release kinetics and optimise device positioning. | Drug-eluting contact lenses or ocular insert. |
| Human factors and patient adherence | Experienced human factors and industrial design team to design intuitive and effective device features. |
Eye drop applicator with guided volume control for elderly patients. |
| Clinician variability for skilled procedures | Experienced human factors and industrial design team to develop ergonomic and fail-safe features for consistent use across skill levels. |
A new microinjection device for suprachoroidal injections. |
| Materials selection | Experienced materials scientists can select the right combination for drug formulation, sterilisation, biocompatibility and manufacturing. |
Coating for a drug-eluting ocular insert that maintains controlled release without leaching or degradation. |
| Precision manufacturing | Able to adapt plastic manufacturing processes to novel designs with tight tolerances. | Micro-moulded injector for an intraocular implant. |
| Sterilisation and aseptic processing | GMP-compliant facilities with cleanroom capabilities in the US. |
Aseptic filling of eye ointment applicators. |
| Scale-up and supply chain | Use Sanner’s global manufacturing network to ensure consistent supply, reduce lead times and co-ordinate bespoke parts. |
Manufacturing of new ocular injection devices at commercial scale. |
| Reducing R&D cost and timeline |
Integrated CDMO allowing for smooth transition from design to manufacture. |
Smooth transition from prototype injector to GMP‑manufactured ocular device. |
Table 2: Novel device development challenges and capabilities to approach them.
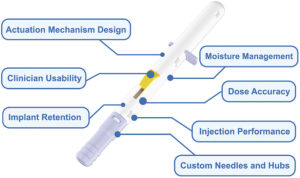
Figure 2: Breakdown of the ocular injection device developed by Sanner.
As an example, Sanner developed a fully customised injection device capable of delivering an extended-release pharmaceutical implant into the vitreous for the treatment of wet AMD and glaucoma (Figures 2 and 3). Drawing on the team’s expertise, Sanner designed a custom needle assembly, including the hub, needle and cap, tailored to the client’s needs. To support early research, Sanner then manufactured a small batch of over a thousand units, enabling critical toxicology testing and a human factors study with key opinion leaders.
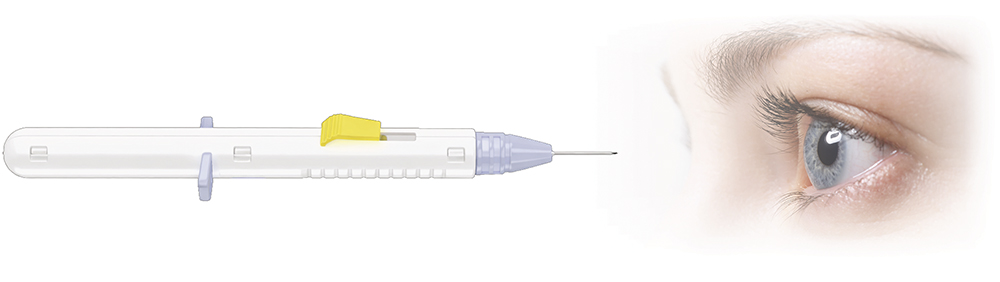
Figure 3: Ocular injection device developed by Sanner for the treatment of wet AMD and glaucoma.
Sanner’s specialised expertise in ophthalmic injection device development supports ocular therapeutics programmes from preclinical stages through to commercialisation. Its team brings experience in designing devices that use a range of active ingredients, including hydrogels. With proven know-how in bringing ophthalmic injectors to market, Sanner serves as a reliable device partner throughout a product’s journey to market success.
Sanner brings decades of experience in medical device engineering, precision manufacturing and pharmaceutical injection moulding in clean room environments (Figure 4). With the right capabilities, Sanner helps customers overcome the many challenges inherent in ophthalmic injector development – accelerating time to market while ensuring safety, performance and regulatory compliance.

Figure 4: Sanner has over 15 years’ experience in manufacturing pipettes for eye drops under cleanroom conditions.
REFERENCES
- Shreya D, Fish PN, Du D, “Navigating the Future of Elderly Healthcare: A Comprehensive Analysis of Aging Populations and Mortality Trends Using National Inpatient Sample (NIS) Data (2010-2024)”. Cureus, 2025, Vol 17(3), art e80442.
- “Global Ocular Drug Delivery Technology Market Size, Share, Growth Analysis Report – Forecast 2034”. Report, Zion Market Research, 2025.
- Santos G et al, “Topical Ocular Drug Delivery: The Impact of Permeation Enhancers. Pharmaceutics”. Pharmaceutics, 2025, Vol 17(4), p 447.
- Egger D et al, “Intravitreal therapy-success stories and challenges”. Wien Med Wochenschr, 2025, Vol 175, pp 162–174.
- Mohan S, Ratra D, “Intravitreal Implants”. StatPearls Publishing, Treasure Island (FL), 2023.
- Gabai A et al, “Innovative Strategies for Drug Delivery to the Ocular Posterior Segment”. Pharmaceutics, 2023, Vol 15(7), art 1862.
- Ranade SV et al, “The Port Delivery System with ranibizumab: a new paradigm for long-acting retinal drug delivery”. Drug Deliv, 2022, Vol 29(1), pp 1326–1334.
- “Roche receives CE mark for Contivue, its Port Delivery Platform containing Susvimo, for neovascular age-related macular degeneration (nAMD)”. Press Release, Roche, Sep 2025.
- “FDA approves Roche’s Susvimo for diabetic retinopathy”. Press Release, Roche, May 2025.
- Peng Y, Tang L, Zhou Y, “Subretinal Injection: A Review on the Novel Route of Therapeutic Delivery for Vitreoretinal Diseases”. Ophthalmic Res, 2017, Vol 58(4), pp 217–226.
- Ladha R et al, “Subretinal Therapy: Technological Solutions to Surgical and Immunological Challenges”. Front Med, 2022, Vol 9, art 846782.
- Irigoyen C et al, “Subretinal Injection Techniques for Retinal Disease: A Review”. J Clin Med, 2022, Vol 11(16), art 4717.
- Kwok JC et al, “Assessment of the Orbit® subretinal delivery system (Orbit® SDS device) and prototypes in adult and juvenile canine eyes”. Drug Deliv and Transl Res, 2025.
- Mi H, MacLaren RE, Cehajic-Kapetanovic J, “Robotising vitreoretinal surgeries”. Eye, 2025, Vol 39, pp 673–682.
- “Scalability of Micro-Intraocular Implants and Devices – Isometric Micro Molding”. Webpage, Nissha Med Tech, accessed Feb 2026.

