To Issue 183
Citation: Cottenden D, Hatton B, “The Retinal Delivery Bottleneck: Why Today’s Tools Won’t Scale”, ONdrugDelivery, Issue 183 (Mar 2026), pp 15–19.
Dr David Cottenden and Benjamin Hatton examine the rapid shift towards cell and gene therapies in ophthalmology, indicated by current pharmaceutical pipelines, and reveal a potential delivery crisis – today’s surgical capacity cannot support what the pipeline demands. Considering this, they explore why a radical rethink of retinal delivery tools could unlock scalable access to the retina.
DRUG DELIVERY TO THE EYE IS CHANGING
The pharmaceutical industry is always in flux, which is the mark of a healthy technology sector. Ophthalmic drugs are no exception – where the landscape was dominated by small molecules 20 years ago, the last 10 to 15 years have seen the rise of biologics and an increasing focus on diseases of the back of the eye. This has, in turn, driven a revolution in workflows to enable an ever-growing number of intravitreal injections to be administered with an astonishing safety and efficacy profile.
“TTP’S ANALYSIS OF PUBLIC DATA ON DRUGS FOR THE EYE PLANNED FOR LAUNCH IN THE US OR EUROPE GIVES AN UNUSUALLY CLEAR ANSWER – THE TREND IN 2035 WILL BE TOWARDS STRONG GROWTH IN CELL AND GENE THERAPIES TARGETING HIGH-PREVALENCE DISEASES OF THE RETINA.”
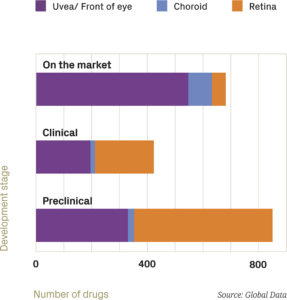
Figure 1: Ophthalmic drugs by target site and development stage.
Given that changes over the last 10 years have been profound, what will drug delivery to the eye look like in ten years from now? TTP’s analysis of public data on drugs for the eye planned for launch in the US or Europe (Figures 1 and 2) gives an unusually clear answer – the trend in 2035 will be towards strong growth in cell and gene therapies targeting high-prevalence diseases of the retina.
Even though many of the drugs in pipelines – especially those in preclinical stages – will not progress to become marketed therapies, drop-out in the drug discovery process is unlikely to change this basic conclusion. Looking first at the target site (Figure 1), while only about 10% of currently marketed therapies target the retina, more than half of the current preclinical assets do. A large majority of the 500 preclinical assets that target the retina will never reach the market, but drugs targeting the retina will represent a much larger proportion of the market than they do today.
Considering the class of therapy (Figure 2), the story is very similar. Currently marketed therapies are mostly small molecules by number (though biologics are more significant by value and, arguably, by impact), whereas they are a minority in the preclinical pipeline, with cell and gene therapies approaching a third of the considered preclinical assets (amounting to more than 250 assets). The trend is clear and robust – so, when these pipeline assets come to fruition, what might the implications for drug delivery be?
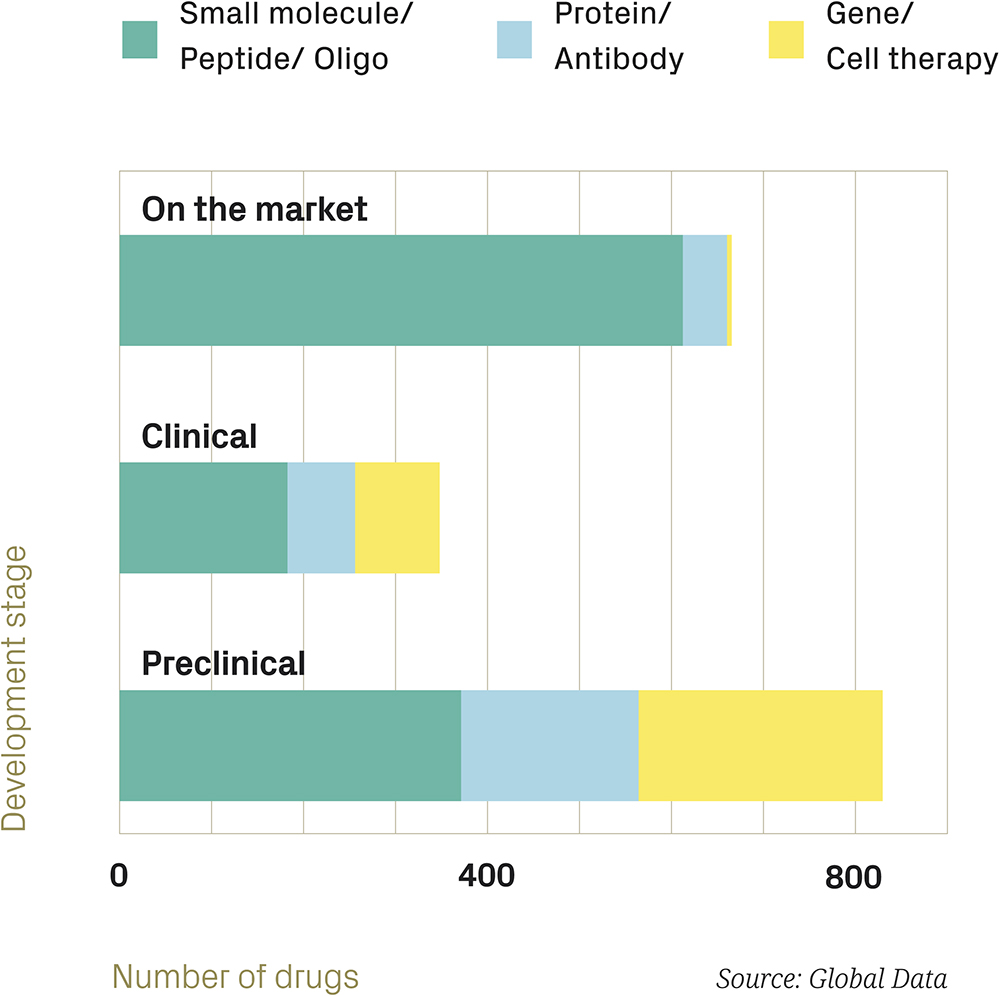
Figure 2: Ophthalmic drugs by drug class and development stage.
IS DELIVERY READY FOR THESE NEW DRUGS?
At present, drug delivery to the eye is dominated by topical eye drops and intravitreal liquid injections. Eye drops are mostly self-administered and provide an effective (although weakly-targeted) method to deliver small molecules to the front of the eye. Intravitreal therapies (IVTs) are administered by healthcare professionals, mostly in non-surgical settings, delivering both small molecules and biologics to the back of the eye. It has been estimated that around 15 million intravitreal injections were performed in the US in 2024.
Can either of these techniques serve the upcoming need to deliver cell and gene therapies to the retina for high-prevalence diseases such as age-related macular degeneration (AMD), diabetic macular oedema (DME) and geographic atrophy (GA)? The answer to this is primarily driven by anatomy – the question is in identifying the structures that exist in the eye between the retina and the delivery site that might limit the spread of cells or gene vectors.
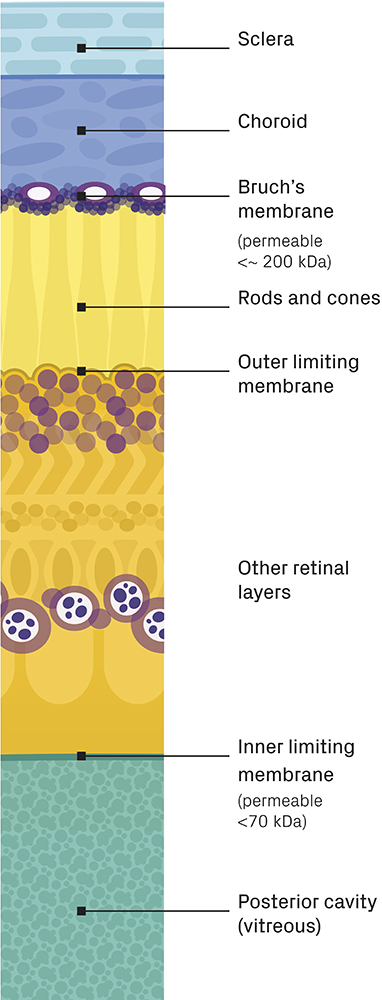
Figure 3: Posterior section of the eye showing sclera, choroid and retinal layers (layer thicknesses are not to scale).
There are essentially two ways into the retina – through the front via the vitreous humour and through the back via the choroid (Figure 3), both of which are regulated by selectively permeable membranes. Bruch’s membrane controls entry via the choroid, only allowing in molecules under approximately 200 kDa (closer to 100 kDa for older patients); whereas the inner limiting membrane controls entry via the vitreous humour, restricting the ability of molecules above approximately 70 kDa to access the retina.
For comparison, a gene therapy with an adeno-associated virus vector is about 5 MDa, and therapeutic cells are, of course, far bigger. Even without considering the effects of outflows from the eye and degradation, it is clear that – exceptional circumstances aside – if cell or gene therapies are delivered outside of the retina, they will stay outside of it.
That said, exceptional circumstances do occur. For example, a cell therapy may treat the retina by introducing cells into the vitreous humour that then manufacture a therapeutic that is small enough to pass into the retina. Furthermore, neither membrane is as simple as a sieve, so slightly larger molecules with appropriate chemical properties will be able to penetrate to some extent. However, the difference in size between the membranes’ “cut-offs” and even a small gene therapy is very large, so delivery outside the retina will often be insufficient.
So, if eye drops and IVTs will not be able to deliver many of the next generation of retinal therapies, what about other existing techniques? There are existing methods for delivering therapies subretinally, of which the more established methods involve accessing the retina from the front via ports in the anterior sclera, typically post-vitrectomy. Another approach (presently still undergoing trials) involves threading a cannula through an incision in the sclera, through the suprachoroidal space and then extending a fine needle into the retina from behind.
Both techniques have a number of limitations. The rate of complications (>50%) and serious adverse events (>1%) is relatively high compared with IVT and, as surgical procedures, they are far more time-consuming (one to two hours versus around 15 minutes) and expensive (over US$10,000 (£7,400) versus around $600).
“EVEN IF THE DOSING REGIMEN FOR NEWER DRUGS WERE TO REDUCE THE NUMBER OF PROCEDURES NEEDED SIGNIFICANTLY, THIS IS NOT A NEAR-MISS – THE CAPACITY OF THE HEALTHCARE SYSTEM TO DELIVER SUBRETINAL DRUGS IS ONLY A FRACTION OF WHAT IS NEEDED.”
However, the biggest limitation by far is availability. There are presently fewer than 20 centres in the US that can deliver these subretinal procedures. But, even if the entire surgical capacity of US retinal specialists were redirected solely to subretinal drug delivery, it would still only enable around 1,500,000 treatments per year. With more realistic constraints, 150,000 treatments per year might be achievable. Even if the dosing regimen for newer drugs were to reduce the number of procedures needed significantly, this is not a near-miss – the capacity of the healthcare system to deliver subretinal drugs is only a fraction of what is needed. Drug delivery to the retina needs a revolution.
HOW MUST DELIVERY DEVICES ADAPT FOR ADVANCED OCULAR THERAPIES?
Having said retinal drug delivery needs a revolution, it is a short step to the need for radically improved delivery tools. The reason is simple – the only way to deal with the essential problem of surgical bandwidth is to reduce the complexity and invasiveness of the delivery procedure to the point where it takes far less time, does not require the same limiting level of skill and, ideally, ceases to be considered a true surgical procedure, much as has already happened with IVT. This level of procedural simplification can be enabled by a complete rethinking of the delivery tools.
So, what does a radically improved delivery tool look like? A lot remains to be determined, but the following key points are likely requirements:
- Direct Delivery to the Retina: This almost certainly means introducing a very fine needle (often 48G) into the retina, probably at an acute angle to the surface to avoid issues with the bevel length.
- Less Invasive: This could mean avoiding the need for vitrectomy and multiple ports in the sclera. The “subretinal-via-the-suprachoroidal-space” approach currently in trials has gone some way towards this.
- Compatible with Cell and Gene Therapies: Amongst other things, this means that the system (perhaps including accessories) must be closed, so as to avoid viral contamination of the environment, and must offer low-shear fluidics to avoid damage to cells.
- De-Skill the Procedure: This is a high bar – and one that varies by territory – but the essence of it is achieving very low infection risk, very low complication rate and a procedure that is both quick and simple.
These requirements are a significant challenge – so is there any reason to think that they might be achievable? For now, a way to consider this is to look at the development of procedures in other areas of surgery. Aortic valve replacement is a good comparison, involving detailed structure, difficult access and high criticality. The evolution of early open-heart procedures, taking five hours with a full sternotomy and cardiopulmonary bypass, into current transcatheter procedures, taking as little as 45 minutes and an incision of less than a centimetre, gives some insight into what might be achievable for retinal delivery.
The example of aortic valve replacement suggests the need to think hard about the following points:
- Access Routes: Identifying a means of accessing the target site by a minor incision in a much less sensitive location, thereby avoiding more traumatic direct access, is key. The exploration of accessing the retina via the suprachoroidal space is promising in this regard, though the challenge of choroidal haemorrhage suggests that there is more still to achieve.
- Effective Imaging: Indirect access always makes visualisation harder, so a combination of imaging techniques will be needed both to guide the clinician to the site and to enable a successful procedure to be performed once it is reached. Ophthalmology is quite well-equipped with imaging approaches, which is a great advantage.
- Robust Stabilisation: “Anchoring” the tools to a solid reference point close to the procedure site is vital for enabling precision and avoiding “jitter”. A good mounting point needs to be solidly connected with the target.
Aortic valve replacement is not a unique example of such profound developments in technique and tooling. While the challenges of retinal drug delivery are substantial, there is every reason to think that they can be met.
IS THERE TIME TO BRIDGE THE GAP?
Pharma pipelines look around ten years into the future – how does that compare with the time that a revolutionary procedure and device will take to come to fruition? Data from TTP’s own extensive product development show that developing a drug delivery device, from a clear vision and a clean sheet through to verification testing, takes four to five years, with scaling and regulatory work fitting around that timescale. However, in this case, where transforming the procedure and creating the product are so interconnected, the device vision is not – and cannot yet be – clear.
In situations like this, it almost never makes sense to begin developing a commercial product straight away, as it will either be too flexible to be economical or easy to use, or be built on assumptions that cannot possibly be evidenced at this stage. Instead, the better approach is usually to create tools to learn with – focused devices that can be used to test approaches and assumptions about core procedures and product direction, including clinician needs and preferences, testing on animal eyes ex vivo, in vivo animal testing, cadaver work and perhaps first-in-human trials.
“COMPARING THESE ACTIVITIES TO TYPICAL PIPELINE TIMESCALES, THERE IS STILL TIME TO THINK BIG AND REVOLUTIONISE DRUG DELIVERY TO THE RETINA – BUT THERE IS LITTLE TIME TO SPARE.”
These tools are designed for ease of iteration and flexibility, not cost and manufacturability; for function and handling, not appearance. They are not an “early prototype” of an eventual product – they are a key means by which the product can be defined. How long this process takes is very context-dependent, but it is usually several years. Comparing these activities to typical pipeline timescales, there is still time to think big and revolutionise drug delivery to the retina – but there is little time to spare.

