Citation: Redding BK, “Overview: Transdermal Delivery Device Design”. ONdrugDelivery Magazine, Issue 49 (May 2014), pp 8-9.
The product development process for a transdermal drug delivery (TDD) system is multidisciplinary in nature. Much of the scientific literature in the field of transdermal delivery pertains to skin permeation and methods of skin penetration enhancement because these are the fundamental issues that must be addressed for any transdermal drug candidate. However, in addition to the basic questions of skin permeability and dose delivered, the development process must also address other basic questions, such as the following:
- What is the appropriate patch design?
- What are the appropriate materials to use in the patch construction?
- Will the target drug be compromised by either the design or the materials used in the patch construction?
- Choice of the skin pathway, either sweat pore, hair follicle, micro-fissure penetration or poration of the skin.
CONVENTIONAL PATCH DESIGNS
The two main traditional/conventional types of patch design – reservoir patches and matrix patches – are shown in figures 1 and 2, respectively.
Reservoir Type Patch
Characterised by the inclusion of a liquid reservoir compartment containing a drug solution or suspension, which is separated from a release liner by a semipermeable membrane and an adhesive.
Commercial examples include:
- Duragesic® (fentanyl, Janssen)
- Estraderm® (estradiol, Novartis (discontinued))
- Transderm-Nitro® (nitroglycerin, Novartis)

Figure 1: Reservoir transdermal patch construction.
Matrix Type Patch
Similar to the reservoir type patch design but has two distinguishing characteristics:
- The drug reservoir is provided within a semisolid formulation
- There is no membrane layer
Commercial examples include:
- Habitol® (nicotine)
- Nitrodisc® (nitroglycerine)
- ProStep® (nicotine)

Figure 2: Drug-in-adhesive matrix patch construction.
Drug-In-Adhesive Type Patch
The drug in adhesive (DIA) patch is a type of matrix match, characterised by the inclusion of the drug directly within the skin-contacting adhesive (Wick, 1988). In this design the adhesive fulfils the adhesion-to-skin function and serves as the formulation foundation, containing the drug and all the excipients (Wilking, 1994). This category also has two sub-sections: monolithic and multilaminate.
Commercial examples include:
- Monolithic DIA: Climara® (estradiol)
- Multilaminate DIA: Nicoderm® (nicotine)
The DIA patch design has several advantages in reducing the size of the overall patch and provides a more concentric seal upon the skin. DIA patches tend to be more comfortable to wear and very thin. A typical DIA patch is 165-200 μm thick.

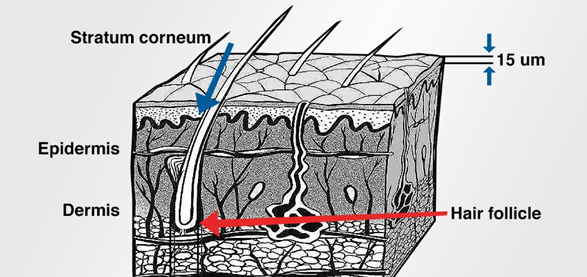
Figure 3: The basic structure of human skin.
Major disadvantages include a longer drug delivery profile. The release of the drug from a DIA patch follows first-order kinetics, that is, it is proportional to the concentration of drug within the adhesive. As the drug is delivered from the DIA patch the drug concentration will eventually begin to fall. The delivery rate therefore falls off over time and this fact needs to be considered in the clinical evaluation phase of development.
A significant problem with most of the main forms of transdermal patch is the intermingling of the drug with adhesive compositions. These result in new profiles and in many instances the drug is degraded through the interaction with the adhesive composition. The chemistry of the adhesive can alter the stability, performance and function of certain drugs. In the case of insulin, for example, the intermingling of the adhesive with the drug can denature the insulin and any deposited insulin would now be a mixture of insulin + adhesive, which may not be a safe blend for dosing purposes.
Additionally there are limits to the molecule size of drugs, which can be delivered via a passive system. Typically drug candidates are below 500 Da for DIA patches and below 1,000 Da for matrix and reservoir patches, even through the use of skin enhancers.
GETTING THROUGH THE SKIN
The skin is a natural barrier, as shown in Figure 3. To deliver a compound transdermally your options are:
- Microporate the skin such as through a catheter or needle. Essentially puncture the skin. Mirconeedle systems and reduced length injectables are designed to reduce the pain associated with skin puncturing.
- Passive absorption through the stratum corneum. Drugs less than 500 Da in size can more easily be absorbed. Drugs between 500 and 1,000 Da require skin enhancers such as alcohol or surfactants to increase skin absorption.
- Dilation of the skin pore. The normal pore diameter is 50 μm (on the body). Dilating the pore to expand its diameter can allow larger molecule drugs to be absorbed.
- Dilation of the pore surrounding the hair follicle. The normal hair follicle pore diameter is 50 μm (on the body). Dilating the pore, to expand its diameter, can allow larger molecules to wick down the hair to the root and from there into the epidermis.
- Fracturing of the skin micro-fissures. By using an energy medium the skin micro-fissures can be dilated and this will allow certain large molecule drugs to be absorbed into the stratum corneum.
- Accelerating the drug to high speeds so it has enough kinetic energy to pass straight through the stratum corneum and other skin layers, coming to rest at the required depth, as with liquid and powder needlefree injection systems.