Citation: Baker C, “Adherence to Self-Medication Can Be Enhanced by Connected Devices”. ONdrugDelivery Magazine, Issue 98 (Jun 2019), pp 30-33.
Craig Baker looks at the factors contributing to non-adherence and explores how connected devices can help patients follow their self-injection regimens.
“Among the technologies proposed to enhance adherence rates, connected devices have shown a great deal of promise.”
A variety of methods of drug delivery, such as autoinjectors, require patients to self-administer medicine as part of their regimen for chronic and acute conditions. Although an increasing number of patients are introduced to these types of devices every year, adherence rates are still less than 50% following six months of treatment.1 The impact of non-adherence on the pharmaceutical industry can include lower product consumption, suboptimal patient outcomes, lower sales, lower expectations of drug efficacy and lower brand equity.1
Among the technologies proposed to enhance adherence rates, connected devices have shown a great deal of promise. These are defined as devices that can be used to capture and transmit information about patient usage, which can subsequently be analysed to gain a better understanding of how patients are using or interacting with their therapies. Noble has recently moved into this space with the development of its AdhereIT™ platform for autoinjector training and injections(Figure 1) to help pharmaceutical companies lower non-adherence rates.The effort to improve adherence rates therefore entails a twofold task. First, it is necessary to understand the nature and scope of non-adherence among patients who self-administer their medicine in general – and via specific devices (e.g. autoinjectors) in particular. This then leads to the second task, which involves focusing on potential solutions—such as new technologies that can be commercialised for the benefit of patients, healthcare providers and pharmaceutical companies.
SEVERAL FACTORS CONTRIBUTE TO NON-ADHERENCE
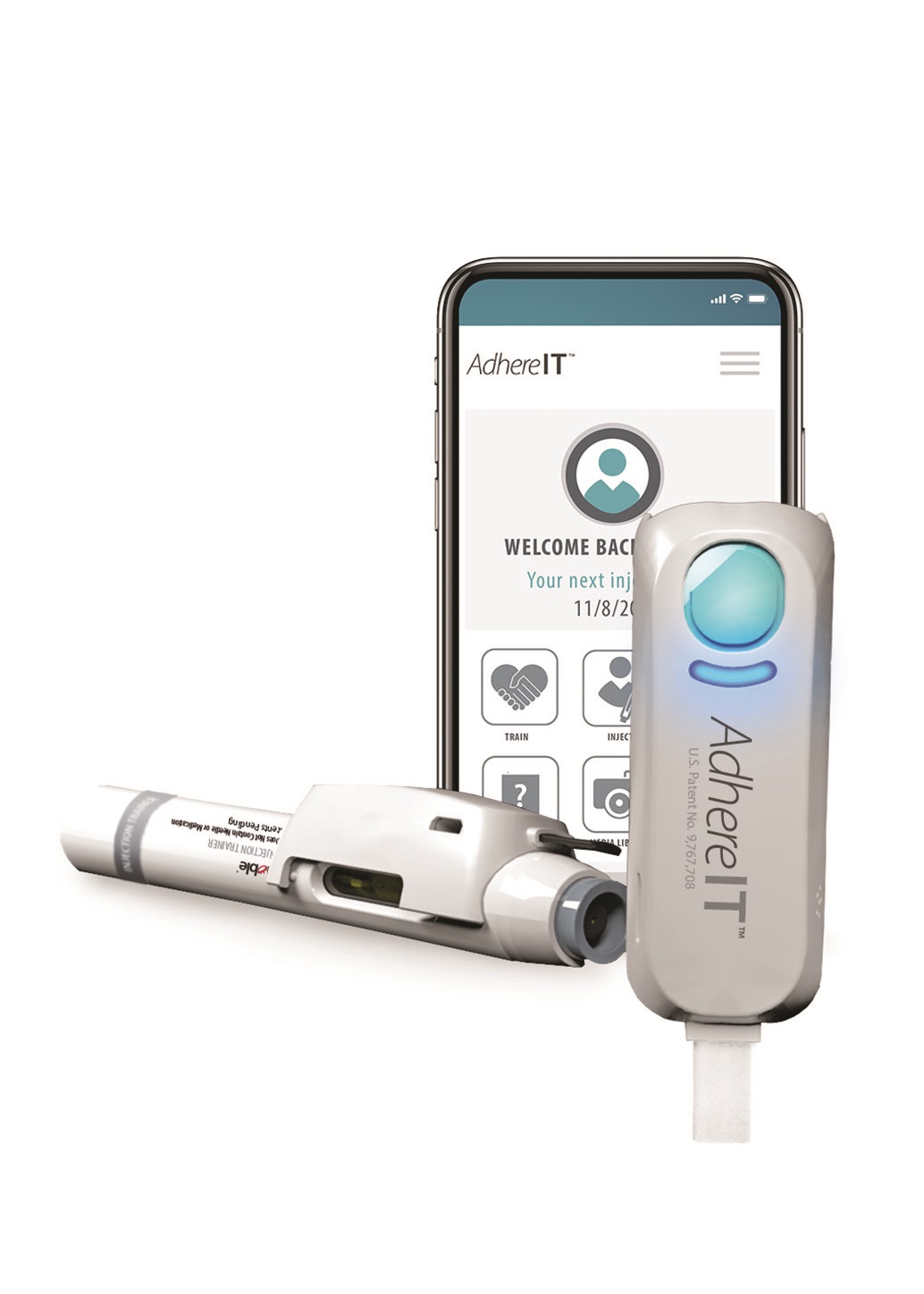
Figure 1: Noble offers customisation of AdhereIT to fit a variety of autoinjector platforms.
There are multiple patient-related factors involved in the problem of non-adherence.2 Some of these are psychological in nature – ranging from simple patient forgetfulness,3 to the fear of being stigmatised by a disease, to patients’ misconceptions regarding the perceived benefit of their treatment. Other reasons patients may not stick with their prescribed course of medication are more practical in nature, such as an inability to pay for a prescription3 or being unable to get the prescription filled, picked up or delivered.3
“According to a study conducted by the University of Texas Medical Branch in Galveston, most patients use their autoinjectors incorrectly.”
Apart from these factors, however, it has also been estimated that 46% of cases of non-adherence result from patients misunderstanding prescription dosing instructions.4 This can result from an unsatisfactory relationship between the healthcare practitioner and the patient, including an inadequate amount of time spent training the patient, less-than-adequate quality of instruction received, an inability to properly understand the instructions for use, and an inadequate appreciation on the healthcare practitioner’s part of the challenges of adherence following the period of initial instruction of the patient on their device (Figure 2).
A review article in The New England Journal of Medicine suggests that a rapid evolution in the time demands placed on healthcare providers is altering how they interact with patients. Specifically, the pressure on physicians to move quickly and accomplish multiple goals during a visit has intensified, and many patients in surveys have described their doctors as relatively hurried and unresponsive. The article suggests various factors have also led to a decrease in face-to-face interactions between doctors and patients. Even as there has been a narrowing in the scope of how doctors interface with patients, there has been a simultaneous growth in team-based instruction of the patient involving other professionals.5 For those charged with training patients on the proper use of autoinjectors, accuracy and professional expertise are clearly vital.
This first 30–90 days after diagnosis – i.e. the onboarding period when the patient is originally introduced and trained on their autoinjector – is crucial for ensuring long-term adherence. However, some studies have shown that patients often have difficulty recalling the exact process of self-administering their medicine with an autoinjector following training. This reflects a study that concluded that 40–80% of all medical information supplied by a healthcare practitioner is forgotten immediately.6 Supplementary to this finding, it has also been suggested that 19% of patients have a higher risk of non-adherence in the wake of poor physician/patient communication.7 When patient training is incomplete or patients recall their training in a faulty manner, the results can include patient error, injury and adverse events – all of which, in turn, may influence patients to curtail use of their device.
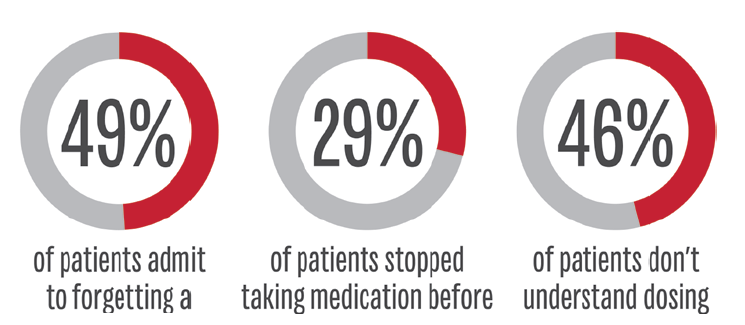
Figure 2: Overview of some the factors contributing to non-adherence.
AUTOINJECTOR USERS PRONETO SPECIFIC ERRORS
Among the subset of patients who specifically use autoinjectors, a common set of errors is likely to play a role. According to a study conducted by the University of Texas Medical Branch in Galveston, most patients use their autoinjectors incorrectly. In the study, patients were asked to demonstrate the steps needed to correctly self-administer their autoinjectors, and it was found that more than half of patients missed three or more steps.8
The mistakes that patients were observed making included: failing to hold the autoinjector in place for the required amount of time; not pressing the device hard enough to trigger the release of the drug; and not choosing a suitable injection site on the body. Despite a redesign of the autoinjector for easier use, most patients continued to make at least one mistake with the device, the study found. In fact, most patients continued to make multiple mistakes.
The most common error, observed among 76% of patients, was a failure to hold the unit in place for at least 10 seconds after triggering. Additional errors involved improper safety cap removal, holding the device in the palm incorrectly, using a swinging motion to place the tip of the autoinjector on the outer thigh and failure to apply sufficient pressure. The poor outcomes resulting from such errors can lead those using autoinjectors to respond negatively to their course of treatment, likely playing a role in inflating their rates of non-adherence.
“AdhereIT can fit onto a trainer that closely mimics an autoinjector as well as onto a prescribed device itself.”
THE ROLE OF CONNECTED DEVICES IN PROMOTING ADHERENCE
To address and alleviate these types of concerns, connected devices have been rising in popularity within the pharmaceutical industry over the past five years. At Noble, an interest in connected devices grew organically out of the company’s role as an industry leader in developing patient-centric advanced drug delivery trainers – including autoinjectors, prefilled syringes, and wearable and respiratory devices. Having studied the errors that were commonly committed during autoinjector training for several years, the company saw an opportunity to help ensure patients could use their prescribed devices properly to sustain adherence.
From this vision, AdhereIT was born. This device offers various categories of features that can make it appealing for patients, healthcare providers and drug companies alike. The first of these features involves its flexible form factor – AdhereIT can fit onto a trainer that closely mimics an autoinjector (Figure 3) as well as onto a prescribed device itself. Drug companies may decide whether to adapt AdhereIT to one or both of these. AdhereIT can also easily fit onto a variety of autoinjectors created by different companies.
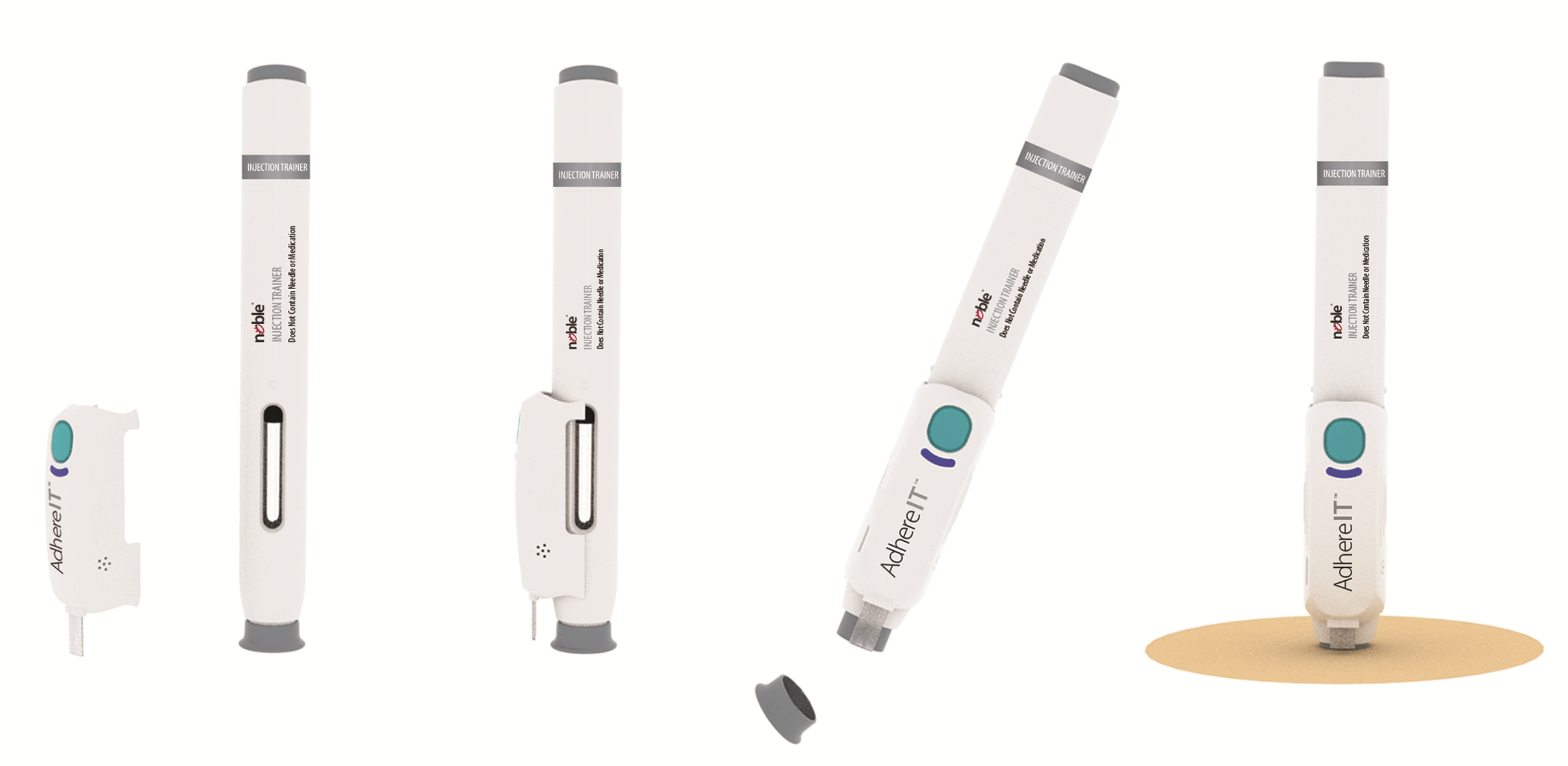
Figure 3: An example of how the AdhereIT adherence device can be used with an autoinjector trainer. It can also fit onto a prescribed device.
The second set of features involves the ability of AdhereIT to detect and monitor how users interact with the specific steps of drug delivery to ensure proper self-injection. For example, the device can detect the precise times at which a training session or an injection with the prescribed device begins and ends, as well as detect when the trainer/device makes contact with the injection site on the skin. It can also send injection scheduling reminders to the patient.
The third set of features involves connectivity. AdhereIT can wirelessly transmit the data it collects regarding the training session or actual injection – including any patient administration errors – to a smartphone or tablet. Its set of high-tech features, in effect, transforms trainers and autoinjectors into “smart” devices. This can provide helpful feedback to the patient in real time during the training period as well as during injection with the prescribed device. AdhereIT has also been configured to integrate with developers’ wireless platforms for enabling the collection and customisation of usage data.
Late last year, Noble received a patent allowance for AdhereIT, and the product was launched into the market soon thereafter. Since then, Noble has continued to refine the platform’s form factors and capabilities.
AdhereIT – and other connected devices like it – provide both short- and long-term benefits for patients, healthcare practitioners and pharmaceutical manufacturers. Most obviously, for patients, the use of these devices may provide the informative feedback that encourages them to adhere to their prescribed dosing regimens for longer periods.
For their part, healthcare practitioners can provide their new autoinjector patients with a tool that provides an added layer of assurance that self-injecting is being performed properly – both during the training phase as well as when the prescribed device is being used. Data collected by the connected device can be shared with the practitioner and reviewed for any irregularities that need to be discussed with the patient.
For drug pharmaceutical manufacturers, the decision to develop a connected device such as AdhereIT for use in tandem with a trainer and prescribed autoinjector can be prudent as well. Specifically, it allows the manufacturer to differentiate itself from competitors in a crowded marketplace and improve the experience and satisfaction of their patients. Noble’s technical expertise and in-house engineering capabilities make the conception, design and manufacturing of the connected device a turnkey process for manufacturers.
Given their potential for enhancing rates of adherence, connected devices such as AdhereIT are likely to play an increasingly important role for patients, their healthcare practitioners and pharmaceutical companies as rates of self-administration rise steadily.
REFERENCES
- Forissier T, “Patient adherence: the next frontier in patient care – Vision & Reality, 9th Edition”. Research Report, Capgemini Consulting, May 26, 2011.
- Bengoa R et al, “Adherence to long-term therapies-evidence in action”. World Health Organization, 2003.
- “The hidden epidemic: Finding a cure for unfilled prescriptions and missed doses”. Boston Consulting Group, November 2003.
- CVS Health, “Adherence: why it’s so hard and what we can do about it”. Insights Report, May 30, 2015.
- Okie S, “The evolving primary care physician”. N Engl J Med, 2012,Vol 366, pp 1849-1853.
- Kessels R, “Patients’ Memory for Medical Information”. J Royal Soc Med, 2003, Vol 96(5), pp 219–222.
- Zolnierekm K, DiMatteo M, “Physician Communication and Patient Adherence to Treatment: A Meta-analysis”. Med Care, 2009, Vol 47(8), pp 826-834.
- Bonds R, Awawa A, Ghazi A, “Misuse of medical devices: a persistent problem in self-management of asthma and allergic disease”. Ann Allergy Asthma Immunol, 2015, Vol 114(1),pp 74-76.e2.


