To Issue 183
Citation: Duband S, Zabala N, “Novelia®: Engineering Precision, Protection and Global Adoption in Preservative-Free Multidose Eye Drop Delivery”, ONdrugDelivery, Issue 183 (Mar 2026), pp 8–13.
Severine Duband and Natalia Zabala discuss the role of multidose preservative-free eyedroppers in the ophthalmic market, highlighting the advantages of Nemera’s Novelia® platform, including its non-return valve system, the sustainability of multidose delivery and the platform’s growing success within the Chinese market.
ENGINEERING MICROBIOLOGICAL INTEGRITY IN PRESERVATIVE-FREE MULTIDOSE DELIVERY
Most ophthalmic eye drops still rely on preservatives to maintain sterility. Benzalkonium chloride, the most widely used preservative, has long been associated with ocular surface toxicity, particularly in chronic conditions such as glaucoma and dry eye disease.1 As treatment durations increase and patients remain on therapy for longer periods, the tolerability of preserved formulations has come under growing scrutiny.
Advances in primary container closure systems have enabled the development of multidose preservative-free (MDPF) solutions designed to maintain sterility without chemical preservatives, while continuing to support ocular surface health and long-term treatment sustainability.2 However, while more than half of currently available MDPF bottles rely on 0.22 μm sterile mesh filters to prevent microbial ingress and manage air compensation, research has questioned the robustness of filter-based systems under real-world conditions.3 Because these filters are porous by design, they do not provide a continuous physical barrier, potentially introducing vulnerabilities during extended in-use periods and repeated patient handling.
Nemera’s alternative approach is based on a non-return valve system combined with a dedicated silicone air-compensation membrane. The non-return valve ensures that no contaminated liquid can be reintroduced into the container after a drop has been dispensed, completely removing the need to filter the liquid pathway. Air intake occurs via a separate venting system that incorporates PureFlow® technology – a solid, homogenous silicone membrane. Unlike bacterial mesh filters, this material is non-porous and contains no holes, allowing its permeability characteristics to be precisely engineered while maintaining a continuous physical barrier against contamination.
An independent review of MDPF systems concluded that Novelia® provides “the largest amount of published information regarding the safety and sterility of these MDPF packages” and demonstrated resilience under both expected real-world microbial challenges and more severe stress scenarios.4
NOVELIA®: DESIGNING FOR DOSE PRECISION AND PATIENT CONTROL
In 2020, Nemera’s R&D department, the Insight Innovation Centre, conducted a qualitative study involving patients and healthcare professionals to explore real-world administration challenges in dry eye and glaucoma therapies, in which interviews consistently revealed a perceived lack of control during eye drop administration. Both dry eye and glaucoma patients highlighted difficulty in reliably delivering a single drop as a primary frustration. Participants reported that multiple drops could be expelled unintentionally or that leakage could occur before actuation. These issues became increasingly pronounced towards the end of a product’s life, when higher squeeze force was required and patients felt progressively less confident in controlling dose delivery.
“CORRECT INSTILLATION REMAINS A SIGNIFICANT BARRIER IN CHRONIC OPHTHALMOLOGY, WITH STUDIES INDICATING THAT A SUBSTANTIAL PROPORTION OF GLAUCOMA PATIENTS STRUGGLE WITH PROPER ADMINISTRATION TECHNIQUE.”
While these challenges may represent inconvenience for some dry eye patients, they are more consequential for glaucoma patients, who may question whether the correct therapeutic dose has been administered and worry about costly medication being depleted prematurely. Correct instillation remains a significant barrier in chronic ophthalmology, with studies indicating that a substantial proportion of glaucoma patients struggle with proper administration technique.1 Therefore, an intuitive, easy-to-use delivery system plays a critical role in supporting adherence, reinforcing patient confidence and ensuring long-term treatment continuity.
Novelia®’s PureFlow® technology not only functions as an air-compensation system but also as a controlled flow mechanism (Figure 1). The integrated design avoids uncontrolled multidrop delivery and supports the consistent release of a single calibrated drop at each actuation, regardless of bottle fill level.
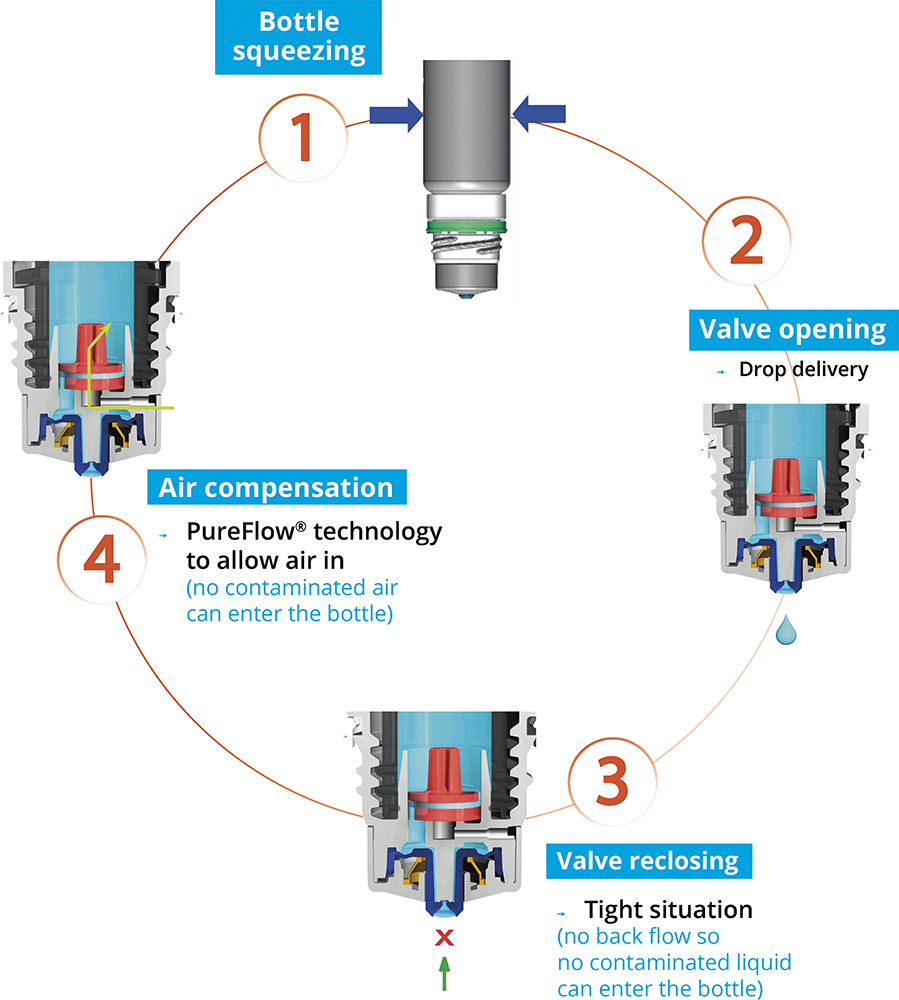
Figure 1: The Novelia® system uses a non-return valve that removes the need to filter the liquid.
To accommodate formulation diversity, Nemera offers three PureFlow® variants engineered for a wide viscosity range, from highly fluid to highly viscous solutions. In addition, five valve sizes are available, enabling precise adjustment of drop size according to product specifications and therapeutic requirements. This modular architecture allows pharmaceutical partners to tailor device performance to the needs of their formulations while enhancing patient confidence, reducing frustration and limiting unnecessary medication waste.
As ophthalmic pipelines increasingly incorporate higher-viscosity systems designed to improve ocular residence time and therapeutic efficacy, delivery platforms must maintain precision, microbiological integrity and usability across diverse formulation profiles. For Nemera, “We Put Patients First” is more than a corporate statement; it reflects a structured and systematic development philosophy. Integrating patient insights early in the design phase enables behavioural considerations to be incorporated into product specifications, risk management processes and user-focused failure mode and effects analysis. By embedding patient needs and practical constraints into formal quality frameworks, Nemera ensures that usability is addressed alongside microbiological safety and mechanical performance.
ERGONOMICS, ACTUATION FORCE AND REAL-WORLD USABILITY
Clinical studies have demonstrated statistically significant variability in the force required to expel a drop from commonly prescribed glaucoma medications.5 Variations in actuation force influence patient confidence, dosing consistency and overall usability, particularly in elderly populations or in patients with reduced dexterity.
In 2015, Nemera commissioned independent user testing to assess comparative device performance and patient perception in real-world settings. Participants were selected based on demographic criteria (age and gender) as well as ophthalmic condition, including glaucoma and dry eye disease. Interviews were conducted in patients’ homes in both the US and the UK to reflect actual usage environments. The study concluded that 76% of participants expressed a preference for Novelia® compared with other MDPF systems available at the time.
“NOVELIA® DEMONSTRATED ONLY A 6% INCREASE IN REQUIRED ACTUATION FORCE FROM THE BEGINNING TO THE END OF TREATMENT, COMPARED WITH APPROXIMATELY 35% OBSERVED IN ALTERNATIVE MDPF SYSTEMS.”

Figure 2: Systane™ Hydration PF MDPF Lubricant Eye Drops for the treatment of dry eye launched in the US market with Novelia® (image courtesy of Alcon, Geneva, Switzerland).
Key drivers of preference included the intuitiveness of the screw-on cap, the reassurance associated with tactile feedback and the stability of squeeze force throughout product life. Novelia® demonstrated only a 6% increase in required actuation force from the beginning to the end of treatment, compared with approximately 35% observed in alternative MDPF systems. Maintaining consistent squeeze performance is particularly important for patients with reduced dexterity, tremors or arthritis, who must be able to handle and manipulate the device reliably for each administration. One Amazon customer review for Systane™ Hydration PF (Figure 2) reads “I have arthritis in my hands and all the other brands are so hard to nearly not working at all to squeeze out even one drop. This dispenser works with gentle pressure every time. I am so glad to have found them.”
In chronic ophthalmic conditions, usability is not a secondary consideration but a determinant of adherence. Variability in squeeze force, inconsistent drop formation or lack of intuitive handling can lead to dosing uncertainty and reduced persistence. By combining controlled flow architecture with ergonomic stability, Novelia® addresses both the mechanical and behavioural dimensions of eye drop administration.
GLOBAL VALIDATION: REGULATORY MILESTONES AND COMMERCIAL SCALABILITY
Novelia® has achieved significant regulatory and commercial milestones in China. The platform is listed on the Center for Drug Evaluation platform (Figure 3),6 enabling referencing of Nemera’s drug master file within drug product applications and facilitating regulatory integration in the Chinese market. Commercial validation has also been achieved through its integration into Eyesucom® (隐形眼镜润眼液) – a preservative-free lubricant eyedrop launched in partnership with Haohai Qisheng (Shanghai, China). Together, Eyesucom’s regulatory publication and market adoption demonstrate the robustness and scalability of the Novelia® MDPF platform within one of the world’s most tightly regulated ophthalmic environments.

Figure 3: Commercial launch of Eyesucom® in China with the Novelia® MDPF platform.
Globally, Novelia® supports more than 550 prescription and over-the-counter (OTC) references across more than 55 countries spanning Europe, Latin America, North America, Oceania, the Middle East and Asia-Pacific. This broad footprint reflects sustained confidence in non-filter-based MDPF technology across both mature and emerging markets.
Novelia’s scalability is further reinforced through its configurability and manufacturing flexibility. Novelia® is available in low-density polyethylene (LDPE) bottle formats of 5, 7.5, 11 and 15 mL (Figure 4), validated for both gamma and ethylene oxide sterilisation processes. Soft technology optimises squeeze force, while blow-fill-insert-seal technology through Nemera’s partner Rommelag (Sulzbach-Laufen, Germany) expands compatibility with diverse formulation and regulatory requirements. Custom colour options enable brand differentiation and alternative cap technologies – including silver-ion and vented configurations – address specific formulation needs. Together with dual-continent manufacturing capabilities, these configurations position Novelia® as a scalable MDPF solution capable of supporting multinational product strategies from development through commercial launch.
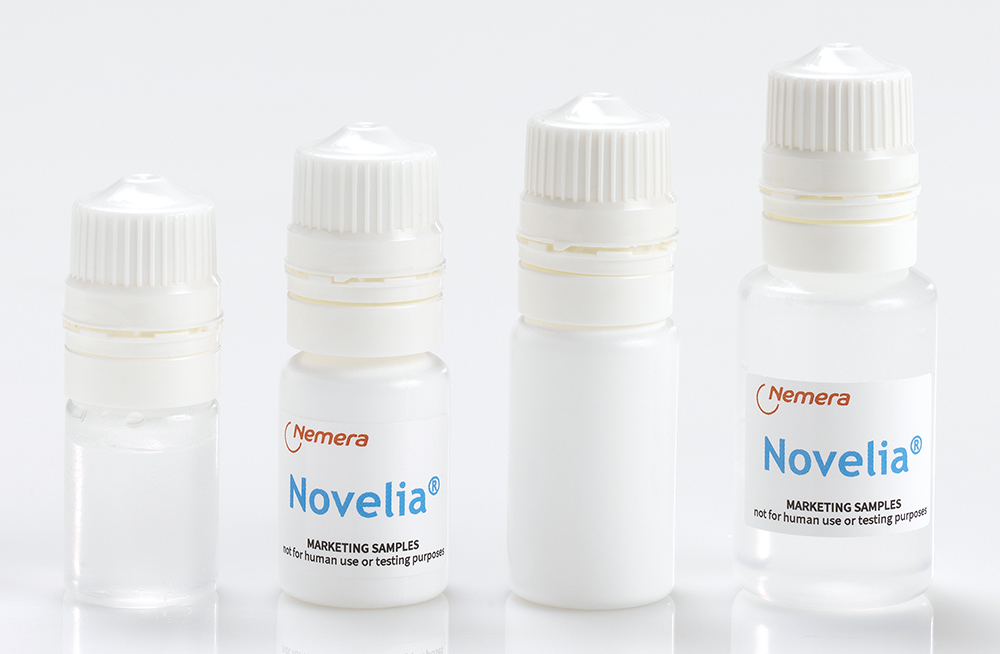
Figure 4: A full range of bottles is available in LDPE, 5, 7.5, 11 and 15 mL.

Figure 5: New ISO 7 cleanroom extension at Nemera’s La Verpillière facility in France, which enables doubled production capacity and reduced lead times.
DUAL-CONTINENT INDUSTRIAL FOOTPRINT: SCALABILITY AND SUPPLY RESILIENCE
To support growing global demand for MDPF systems, Nemera has further expanded its manufacturing capabilities in the US, effectively doubling production capacity for Novelia® multidose eyedroppers (Figure 5). This expansion strengthens the company’s dual-continent manufacturing capabilities and reinforces supply resilience for global pharmaceutical partners. The new ISO 7 cleanroom facility spans more than 9,000 square feet and integrates high-speed assembly and injection-moulding capabilities, enhancing scalability while maintaining the stringent quality standards required for drug-device combination products.
In parallel, Nemera has expanded its capacity at its La Verpillière site in France through the addition of a new production line for Novelia® Standard and Novelia® Vent devices using identical core technology and components. The extension supports larger batch sizes and shorter lead times under established quality control processes. Collectively, these investments strengthen Nemera’s industrial scalability while preserving the high standards expected for combination products.
SUSTAINABILITY IN MDPF: FROM FORMAT EFFICIENCY TO MATERIAL INNOVATION
Single-use unit doses raise concerns regarding cost, waste and convenience, particularly in chronic therapies.4 Handling challenges among elderly patients may also increase contamination risk.7 With the global glaucoma population projected to exceed 111 million by 2040,8 practical and sustainable long-term delivery formats are becoming increasingly important.
Nemera conducted a comparative analysis evaluating Novelia® MDPF delivery against single-use unit-dose packaging for a typical glaucoma regimen (one drop per eye, twice daily) over one month. The multidose configuration used eight times less plastic, generated 25 times less drug waste and required nine times less transportation energy compared with the equivalent unit-dose format, highlighting the environmental advantages of MDPF systems across the treatment lifecycle.
Beyond format efficiency, sustainability also requires addressing material impact. Lifecycle assessment identified raw materials as a significant contributor to a product’s carbon footprint. In response, Nemera developed a bio-based resin version of Novelia®, derived from second-generation crude tall oil residues. This alternative reduces Novelia’s carbon footprint while maintaining identical device design, performance and pharmaceutical-grade compliance. Extractables testing confirmed regulatory compatibility, enabling adoption of the bio-resin configuration without altering device functionality or quality standards.
Nemera’s broader sustainability strategy aligns carbon reduction, responsible sourcing and eco-design objectives, supported by EcoVadis Gold status (top 5% globally) and Science Based Targets initiative commitments across operations and supply chain.
INTEGRATED COMBINATION PRODUCT SUPPORT
Nemera provides laboratory services including in-use simulation over two weeks, drop size analysis (variable depending on valve size), flow control and squeeze-force evaluation to determine the optimal Novelia® configuration for each formulation. Based on performance data, Nemera’s team recommends the appropriate PureFlow® control, bottle type, valve size and cap technology aligned with therapeutic requirements.
These programmes may also include microbiological challenge testing on the combination product, as well as functional assessment under extreme storage conditions, including temperature cycling and stress scenarios. The outcome is submission-ready documentation designed to support global regulatory filings and de-risk development timelines.
“THROUGH A NETWORK OF FORMULATION LICENSORS, FILLING PARTNERS AND INDUSTRIAL COLLABORATORS, NEMERA SUPPORTS THE DEVELOPMENT OF FULLY INTEGRATED DRUG- DEVICE COMBINATION PRODUCTS, ACCELERATING TIME TO MARKET WHILE MAINTAINING COMPLIANCE WITH REGIONAL REGULATORY FRAMEWORKS.”
Nemera’s regulatory experts support customers throughout the submission process, providing guidance on required documentation, technical files and supportive data packages for registration. In addition, Nemera can facilitate access to ready-to-use dossiers for selected molecules suitable for private labelling in combination with the Novelia® delivery system. Through a network of formulation licensors, filling partners and industrial collaborators, Nemera supports the development of fully integrated drug-device combination products, accelerating time to market while maintaining compliance with regional regulatory frameworks.
While device ergonomics play a central role in usability, patient education remains equally important in mitigating unintentional non-compliance.9 Educational resources supporting correct eye drop administration improve technique and adherence. Nemera therefore extends its support beyond device supply, assisting customers during product launch through dedicated training for sales teams and healthcare professionals, as well as materials to support promotional and educational initiatives. Customisable patient guidance videos, available in multiple languages, further reinforce correct usage and adherence across global markets.
THE FUTURE OF MDPF: PERFORMANCE, SUSTAINABILITY AND SCALE
Over the next three to five years, MDPF systems are expected to become standard across both prescription and over-the-counter ophthalmics, driven by long-term ocular surface protection, patient convenience and sustainability. At the same time, formulation innovation is progressing towards higher-viscosity systems and advanced therapeutic approaches, placing greater demands on delivery precision, microbiological integrity and device-formulation compatibility.
In this evolving landscape, MDPF platforms must combine configurability, regulatory robustness, industrial scalability and environmental responsibility. Through continued investment in technology, sustainable materials and global manufacturing capacity, Nemera aims to support the next generation of safe, effective and patient-centric ophthalmic therapies.
REFERENCES
- Baudouin C et al, “Preservatives in eyedrops: the good, the bad and the ugly”. Prog Retin Eye Res, 2010, Vol 29(4), pp 312–334.
- “Ophthalmic Product Development From Bench to Bedside” (Neervannan S, Kompella UB, eds). Springer, 2021, AAPS Vol 37.
- Hasegawa H et al, “Membrane filter (pore size, 0.22-0.45 micro m; thickness, 150 micro m) passing-through activity of Pseudomonas aeruginosa and other bacterial species with indigenous infiltration ability”. FEMS Microbiol Lett, 2003, Vol 223(1), pp 41–46.
- Campolo A, Crary M, Shannon P, “A Review of the Containers Available for Multi-Dose Preservative-Free Eye Drops”. Biomed J Sci Tech Res, 2022, Vol 45(1), pp 36035–36044.
- Moore DB et al, “Squeeze Me if You Can: Variability in Force Requirements to Extract a Drop From Common Glaucoma Bottles”. J Glaucoma, 2016, Vol 25(9), pp 780–784.
- “Novelia®, published on CDE (Center for Drug Evaluation) platform in China!”. Press release, Nemera, Oct 2022.
- Bagnis A et al, “Antiglaucoma drugs: The role of preservative-free formulations”. Saudi J Ophthalmol, 2011, Vol 25(4), pp 389–394.
- Tham Y-C et al, “Global prevalence of glaucoma and projections of glaucoma burden through 2040: a systematic review and meta-analysis”. Ophthalmology, 2014, Vol 121(11), pp 2081–2090.
- Davis SA et al, “A randomized controlled trial of an online educational video intervention to improve glaucoma eye drop technique”. Patient Educ Couns, 2019, Vol 102(5), pp 937–943.

