To Issue 183
Citation: Kalitzki A, Wyler S, Aksel S, “SHL Medical and Bayer: A Collaborative Approach to Advancing Intravitreal Paediatric Dosing”, ONdrugDelivery, Issue 183 (Mar 2026), pp 24–29.
Andreas Kalitzki, Samuel Wyler and Dr Seda Aksel explain how Bayer and SHL Medical collaborative efforts are enhancing paediatric intravitreal injections through evidence-based dosing strategies and clinically validated therapies using SHL Medical’s Micro Dosing Device technology.
“THE CLINICAL USE OF IVTs HAS SEEN STEADY GROWTH, PARTICULARLY IN RESPONSE TO RISING RATES OF SIGHT THREATENING CONDITIONS SUCH AS AGE-RELATED MACULAR DEGENERATION, DIABETIC RETINOPATHY AND ROP.”
The clinical use of intravitreal therapies (IVTs) has seen steady growth, particularly in response to rising rates of sight-threatening conditions such as age-related macular degeneration, diabetic retinopathy and retinopathy of prematurity (ROP). These conditions often require localised injections directly into the vitreous body of the eye, where the precision of both the drug and its delivery play a central role in patient outcomes.
ROP is a disease of retinal vascular development that occurs in premature infants. It is characterised by the abnormal growth of immature retinal blood vessels, which can progress to fibrovascular proliferation, retinal detachment and ultimately blindness if untreated.1
Low birth weight under 1,500 g (3.3 lb) and early gestational age (<32 weeks) are the major risk factors for ROP, making it one of the leading causes of childhood blindness worldwide. In the UK, ROP screening is performed in preterm infants who meet these risk criteria. A 2011 UK cohort study reported an incidence of 12.6% in this population, and rates have continued to rise globally as the survival of smaller preterm infants improves.2–4
ROP is a complex retinal vascular disease, often developing in two overlapping phases:
- Phase I (Hyperoxia-Induced Vessel Cessation): Preterm infants often receive supplemental oxygen, which halts the normal development of retinal blood vessels by suppressing angiogenesis driven by vascular endothelial growth factor (VEGF). This leads to large areas of avascular peripheral retina.
- Phase II (Hypoxia-Induced Pathological Neovascularisation): As the retina matures, it becomes hypoxic in those avascular zones. The resulting surge in VEGF triggers the growth of fragile, disorganised new blood vessels. These vessels may leak, form fibrovascular membranes and cause retinal traction or detachment.
If left untreated, the advanced stages of ROP can lead to retinal detachment and lifelong vision loss (Figure 1).

Figure 1: (A) Normal vision and (B) vision affected by macular degeneration. Image courtesy of the National Eye Institute, National Institutes of Health (Public domain).
ROP severity is classified according to the International Classification of ROP (ICROP). These diagnostic categories classify ROP based on:
- Location (Zone): Zones (I–III) describe the anatomical location of disease within the retina. This is illustrated in Figure 2A.
- Severity (Stage): Stages (1–5) reflect the severity of retinal changes, ranging from Stage 1 (a demarcation line) to Stage 5 (total retinal detachment). This is illustrated in Figure 2B.
- Posterior Pole Vascular Features (Normal, Pre-Plus or Plus Disease): Plus disease is a hallmark of aggressive ROP, where retinal vessels show marked dilation and tortuosity. It is a sign of worsening pathology and indicates a high risk of progression to more severe ROP. Its presence often signals the need for immediate treatment.5,6
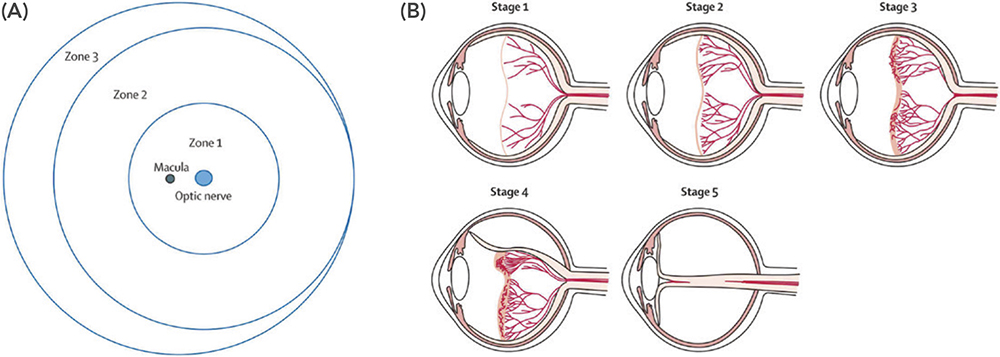
Figure 2: Zones and progressive stages of ROP. Figure A shows that the retina is divided into three zones. Figure B illustrates the classification of ROP according to stages: Stage 1 ROP – A faint demarcation line marks the boundary between vascularised and avascular retina; Stage 2 ROP – The line becomes a ridge, indicating abnormal vascular proliferation; Stage 3 ROP – Extraretinal fibrovascular proliferation extends into the vitreous, increasing the risk of retinal detachment; Stage 4 ROP – Partial retinal detachment; and Stage 5 ROP – Total retinal detachment. Reproduced from Retinopathy of Prematurity—A Brief Review by Saba Al Rashaed, licensed under CC BY 4.0.
EYLEA® (aflibercept – Regeneron Pharmaceuticals, Tarrytown, NY, US), a recombinant fusion protein designed to bind VEGF-A, VEGF-B and placental growth factor, has established its role as an effective and targeted therapy for ROP. Its ability to inhibit multiple angiogenic factors makes it particularly suitable for severe or aggressive cases of ROP. Importantly, its use in this indication is gaining traction following results from randomised clinical trials, such as the Phase III study FIREFLEYE, which demonstrated encouraging regression rates in infants with treatment-requiring ROP.7
In the treatment of ROP, especially with anti-VEGF agents such as aflibercept, the precision of the administered dose is critical – both for therapeutic success and to minimise risk. The recommended intravitreal dose for ROP is 0.4 mg per eye, corresponding to a volume of 10 µL. Delivering a precise microlitre-scale volume is inherently challenging, and small deviations can lead to under-treatment (risking recurrence) or over-treatment (increasing intraocular pressure and systemic complications).
Data from ongoing trials and real-world case series show that recurrence of ROP is significantly reduced when aflibercept is delivered at the appropriate dosage with strict control over injection volume and technique. For example, in the FIREFLEYE study, treatment with aflibercept 0.4 mg led to high regression rates and minimal systemic VEGF suppression – provided the injection volume was carefully calibrated.7
Studies have also shown that administration with syringes – such as 1 mL disposable syringes or glass prefilled syringes (PFSs) – often exhibit significant variability when delivering microlitre doses. This variability becomes clinically significant when the therapeutic margin is narrow, as is the case with aflibercept in neonatal ROP. Developed by SHL Medical in partnership with Bayer, Picleo® is helping to deliver the precise volume for the paediatric preterm indication, without significantly changing the IVT process in terms of injection workflow.8,9
“THE PICLEO® PAEDIATRIC DOSING DEVICE IS SPECIFICALLY DESIGNED TO ENSURE SAFE AND RELIABLE INTRAVITREAL DELIVERY OF EYLEA IN PRETERM INFANTS.”
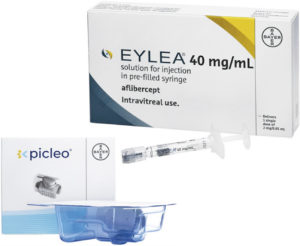
Figure 3: Image inset of EYLEA® (aflibercept) and the Picleo® paediatric dosing device. The device enables high dosing accuracy volumes as low as 10 μL.
THE PICLEO® PAEDIATRIC DOSING DEVICE
The Picleo® paediatric dosing device is specifically designed to ensure safe and reliable intravitreal delivery of EYLEA in preterm infants (Figure 3). Developed based on SHL Medical’s Micro Dosing Device (MDD) technology, the device is compatible with standard Luer lock PFSs and needles via Luer lock interfaces. Key features of the device include:
- Fixed-Dose Volume Delivery: Engineered to administer a precise nominal dose – drastically reducing variability seen with conventional syringes10
- PFS Compatibility: Designed to directly accommodate Bayer’s EYLEA® (aflibercept 40 mg/mL) PFS with Luer lock connection and a 30G needle, ensuring a closed, controlled system during preparation
- Ease of Preparation and Safety: The device includes a visual window for priming, explicit instructions to eliminate air bubbles (which are a potential cause of underdosing) and ergonomic grips to hold the device, preventing accidental actuation
- Ease of Administration: One-click dosing mechanism – the dose button delivers the fixed volume, with no need for dose mark alignment, eliminating human errors in microlitre dosing
- Regulatory Compliance: Compliant with EU Medical Device Regulation (MDR 2017/745) and intended for ROP administration; provided sterile for single use to reduce the risk of infection.

Figure 4: SHL Medical’s MDD technology and PFS.
The MDD technology (Figure 4) developed by SHL Medical is designed to enable the accurate, repeatable delivery of fixed microlitre-scale drug volumes. This is independent of user technique, addressing the inherent limitations of conventional syringes at very low doses. To use the MDD, it is first attached between the PFS and an injection needle. The plunger rod is slowly depressed to prime the system, expelling any air and filling the chamber within the device. The push button is subsequently used to mechanically deliver the controlled volume, thereby minimising variability associated with applied hand force, visual dose alignment and syringe tolerances. An overview of the specifications of the device can be seen in Table 1.
| Specification | SHL & Bayer’s Picleo Device |
| Device Type | MDD |
| Primary Container | Disposable or PFS with standard Luer lock connection |
| Usage | Single fixed-dose, disposable |
| Needle Attachment | Manual, compatible with conventional hypodermic needles with female Luer lock connector |
| Syringe Attachment | Manual, compatible with syringes with male Luer lock connector |
| Route of Administration | Subcutaneous, intravenous and intravitreal |
| Injection Feedback | Audible click at the end-of-dose, visual and tactile feedback |
Table 1: Technical specifications of SHL Medical’s MDD technology.
DOSING PERFORMANCE EVALUATION
A comparative analysis was conducted to assess the dosing accuracy of the MDD in combination with a PFS versus a disposable syringe with a visual indicator to establish a 10 µL dose. Both delivery systems were filled with aflibercept (40 mg/mL) and used a 30G x ½” injection needle. The study focused on key metrics such as dose accuracy and repeatability.
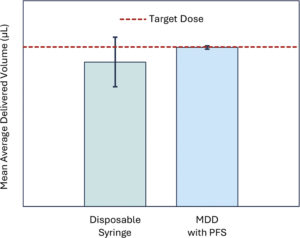
The baseline dataset generated with the conventional disposable 1 mL syringe showed a high standard deviation of 2.09 µL, indicating substantial user-dependent variability and poor task-to-task consistency. In contrast, the dataset obtained with the MDD exhibited a mean value closely aligned with the target and a markedly lower standard deviation of 0.13 µL. The higher accuracy, improved repeatability and superior procedural control is illustrated by the difference between the error bars shown in Figure 5.
The results from a statistical comparison using a two-sample t-test (t = –4.65, p < 0.0001) confirmed that the performance difference was significant. These findings provide strong evidence that the MDD offers substantial usability advantages over the conventional syringe method. The marked reduction in variability and improved alignment to target values indicate that the MDD’s design enables more intuitive operation, finer dose control and a lower likelihood of user error.
FORMATIVE USABILITY TESTING
A usability study (Figure 6) was conducted to evaluate the safe and effective use of the Picleo® paediatric dosing device for administering IVTs in premature infants with ROP. The study involved six experienced ophthalmologists in Germany and was designed as a face-to-face, simulated-use study that replicated a neonatal intensive care unit or operating room environment. Each participant performed a full injection workflow using a placebo-filled PFS and a 30G x ½” injection needle, simulating administration into dummy eyes mounted on a model infant. The procedure included unpacking the product, reviewing the instructions for use, assembling and priming the system, and performing a simulated injection. Hand size and grip strength were also measured to assess ergonomic factors. The results of this study confirmed the overall comprehensibility, adequacy and acceptability of the system and its accompanying instructions. The usability evaluation provided objective evidence that the device can be used safely and effectively by the intended users.
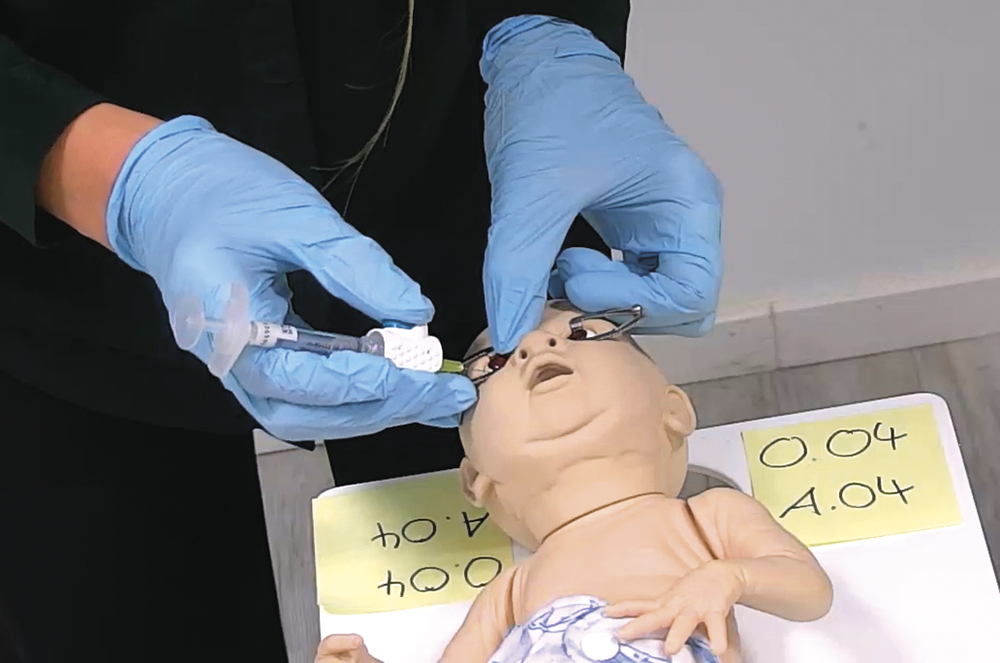
Figure 6: Handling technique during a simulated injection with the Picleo paediatric dosing device.
“THE SUCCESSFUL TREATMENT OF ROP HINGES ON ACHIEVING A DELICATE BALANCE: DELIVERING AN EFFECTIVE DOSE WHILE MINIMISING OCULAR AND SYSTEMIC RISK IN AN EXTREMELY VULNERABLE PATIENT POPULATION.”
CONCLUSION
The successful treatment of ROP hinges on achieving a delicate balance: delivering an effective dose while minimising ocular and systemic risk in an extremely vulnerable patient population. Bayer’s continued efforts in paediatric retinal disease reflect a clear commitment to addressing this unmet medical need through evidence-based dosing strategies and clinically validated therapies such as aflibercept. By enabling reliable administration of the recommended low dose and small injection volume without fundamentally altering the established IVT workflow, Bayer supports both therapeutic precision and clinical practicality – two essential factors for adoption in neonatal care settings.
At the same time, SHL Medical’s MDD technology represents a broader vision for precision drug delivery beyond intravitreal use. Designed as a flexible solution, the MDD has the potential to add value across multiple therapeutic areas where fixed, low-volume dosing is critical and conventional delivery systems fall short. Collaboration between device companies and pharmaceutical partners can bridge the gap between therapies and those who need them, enabling safer, more precise dosing and improved patient outcomes.
REFERENCES
- Kim SJ et al, “Retinopathy of prematurity: a review of risk factors and their clinical significance”. Surv Ophthalmol, 2018, Vol 63(5), pp 618–637.
- Sen P et al, “Retinopathy of prematurity treatment: Asian perspectives”. Eye (Lond), 2020, Vol 34 (4), pp 632–642.
- Hong EH, Shin YU, Cho H, “Retinopathy of prematurity: a review of epidemiology and current treatment strategies”. Clin Exp Pediatr, 2022, Vol 65(3), pp 115–126.
- Dou G-R et al “Demographic profile and ocular characteristics of stage 5 retinopathy of prematurity at a referral center in Northwest China: implications for implementation”. BMC Ophthalmol, 2018, Vol 18(1), art 307.
- Chiang MF et al, “International Classification of Retinopathy of Prematurity, Third Edition”. Ophthalmology, 2021, Vol 128(10), pp e51–e68.
- Al Rashaed S, “Retinopathy of Prematurity— A Brief Review.” Dr. Sulaiman Al Habib Medical Journal, 2019, Vol 1(3–4), pp 58–64.
- Stahl A et al, “Systemic exposure to aflibercept after intravitreal injection in premature neonates with retinopathy of prematurity: results from the FIREFLEYE randomized phase 3 study”. Eye (Lond), 2024, Vol 38(8), pp 1444–1453.
- Moisseiev E et al, “Effect of Syringe Design on the Accuracy and Precision of Intravitreal Injections of Anti-VEGF Agents”. Curr Eye Res, 2017, Vol 42(7), pp 1059–1063.
- Ekinci DY, Vural AD, “Comparison of two different doses of intravitreal aflibercept in the treatment of retinopathy of prematurity”. J AAPOS, 2021, Vol 25 (2), pp 93.e1–93.e5.
- “Instructions for Use: Picleo® Paediatric Dosing Device for the administration of 10 µL nominal dose of Eylea® in pre-filled syringe in ROP”. Bayer, 2023.

